VEGFR2 promotes central endothelial activation and the spread of pain in inflammatory arthritis
- PMID: 29548992
- PMCID: PMC6302073
- DOI: 10.1016/j.bbi.2018.03.012
VEGFR2 promotes central endothelial activation and the spread of pain in inflammatory arthritis
Abstract
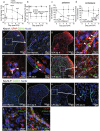
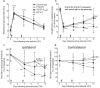
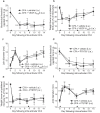
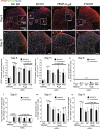
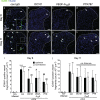
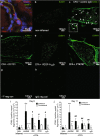
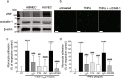
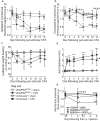
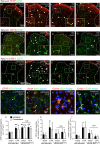
Chronic pain can develop in response to conditions such as inflammatory arthritis. The central mechanisms underlying the development and maintenance of chronic pain in humans are not well elucidated although there is evidence for a role of microglia and astrocytes. However in pre-clinical models of pain, including models of inflammatory arthritis, there is a wealth of evidence indicating roles for pathological glial reactivity within the CNS. In the spinal dorsal horn of rats with painful inflammatory arthritis we found both a significant increase in CD11b+ microglia-like cells and GFAP+ astrocytes associated with blood vessels, and the number of activated blood vessels expressing the adhesion molecule ICAM-1, indicating potential glio-vascular activation. Using pharmacological interventions targeting VEGFR2 in arthritic rats, to inhibit endothelial cell activation, the number of dorsal horn ICAM-1+ blood vessels, CD11b+ microglia and the development of secondary mechanical allodynia, an indicator of central sensitization, were all prevented. Targeting endothelial VEGFR2 by inducible Tie2-specific VEGFR2 knock-out also prevented secondary allodynia in mice and glio-vascular activation in the dorsal horn in response to inflammatory arthritis. Inhibition of VEGFR2 in vitro significantly blocked ICAM-1-dependent monocyte adhesion to brain microvascular endothelial cells, when stimulated with inflammatory mediators TNF-α and VEGF-A165a. Taken together our findings suggest that a novel VEGFR2-mediated spinal cord glio-vascular mechanism may promote peripheral CD11b+ circulating cell transmigration into the CNS parenchyma and contribute to the development of chronic pain in inflammatory arthritis. We hypothesise that preventing this glio-vascular activation and circulating cell translocation into the spinal cord could be a new therapeutic strategy for pain caused by rheumatoid arthritis.
Keywords: CD11b; Chronic pain; Glio-vascular activation; ICAM-1; Inflammatory pain; Mechanical allodynia; Microglia, mono-arthritis; Rheumatoid arthritis; VEGFR2.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Figures

References
-
- Abram S.E., Yi J., Fuchs A. Permeability of injured and intact peripheral nerves and dorsal root ganglia. Anesthesiology. 2006;105(1):146–153. - PubMed
-
- Andrew D., Greenspan J.D. Mechanical and heat sensitization of cutaneous nociceptors after peripheral inflammation in the rat. J. Neurophysiol. 1999;82(5):2649–2656. - PubMed
-
- Anzil A.P., Blinzinger K., Herrlinger H. Fenestrated blood capillaries in rat cranio-spinal sensory ganglia. Cell Tissue Res. 1976;167(4):563–567. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

