Mechanical stretch induced transcriptomic profiles in cardiac myocytes
- PMID: 29549296
- PMCID: PMC5856749
- DOI: 10.1038/s41598-018-23042-w
Mechanical stretch induced transcriptomic profiles in cardiac myocytes
Abstract
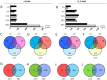
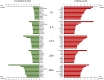
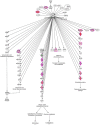
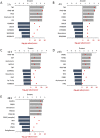
Mechanical forces are able to activate hypertrophic growth of cardiomyocytes in the overloaded myocardium. However, the transcriptional profiles triggered by mechanical stretch in cardiac myocytes are not fully understood. Here, we performed the first genome-wide time series study of gene expression changes in stretched cultured neonatal rat ventricular myocytes (NRVM)s, resulting in 205, 579, 737, 621, and 1542 differentially expressed (>2-fold, P < 0.05) genes in response to 1, 4, 12, 24, and 48 hours of cyclic mechanical stretch. We used Ingenuity Pathway Analysis to predict functional pathways and upstream regulators of differentially expressed genes in order to identify regulatory networks that may lead to mechanical stretch induced hypertrophic growth of cardiomyocytes. We also performed micro (miRNA) expression profiling of stretched NRVMs, and identified that a total of 8 and 87 miRNAs were significantly (P < 0.05) altered by 1-12 and 24-48 hours of mechanical stretch, respectively. Finally, through integration of miRNA and mRNA data, we predicted the miRNAs that regulate mRNAs potentially leading to the hypertrophic growth induced by mechanical stretch. These analyses predicted nuclear factor-like 2 (Nrf2) and interferon regulatory transcription factors as well as the let-7 family of miRNAs as playing roles in the regulation of stretch-regulated genes in cardiomyocytes.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Mechanical stretch via transforming growth factor-β1 activates microRNA-208a to regulate hypertrophy in cultured rat cardiac myocytes.J Formos Med Assoc. 2013 Oct;112(10):635-43. doi: 10.1016/j.jfma.2013.01.002. Epub 2013 Feb 4. J Formos Med Assoc. 2013. PMID: 24120154
-
Stretch-induced hypertrophy activates NFkB-mediated VEGF secretion in adult cardiomyocytes.PLoS One. 2011;6(12):e29055. doi: 10.1371/journal.pone.0029055. Epub 2011 Dec 13. PLoS One. 2011. PMID: 22174951 Free PMC article.
-
Mechanical stretch induces endothelial nitric oxide synthase gene expression in neonatal rat cardiomyocytes.Clin Exp Pharmacol Physiol. 2009 May;36(5-6):559-66. doi: 10.1111/j.1440-1681.2008.05100.x. Epub 2008 Oct 28. Clin Exp Pharmacol Physiol. 2009. PMID: 19673940
-
Effect of atorvastatin on cardiomyocyte hypertrophy through suppressing MURC induced by volume overload and cyclic stretch.J Cell Mol Med. 2019 Feb;23(2):1406-1414. doi: 10.1111/jcmm.14044. Epub 2018 Dec 3. J Cell Mol Med. 2019. PMID: 30511410 Free PMC article.
-
Role of the renin-angiotensin system in cardiac hypertrophy.Am J Cardiol. 1999 Jun 17;83(12A):53H-57H. doi: 10.1016/s0002-9149(99)00259-3. Am J Cardiol. 1999. PMID: 10750588 Review.
Cited by
-
ChGn-2 Plays a Cardioprotective Role in Heart Failure Caused by Acute Pressure Overload.J Am Heart Assoc. 2022 Apr 5;11(7):e023401. doi: 10.1161/JAHA.121.023401. Epub 2022 Mar 24. J Am Heart Assoc. 2022. PMID: 35322673 Free PMC article.
-
Mechanotransduction Circuits in Human Pathobiology.Int J Mol Sci. 2024 Mar 29;25(7):3816. doi: 10.3390/ijms25073816. Int J Mol Sci. 2024. PMID: 38612628 Free PMC article.
-
The ion channel Trpc6a regulates the cardiomyocyte regenerative response to mechanical stretch.Front Cardiovasc Med. 2024 Jan 8;10:1186086. doi: 10.3389/fcvm.2023.1186086. eCollection 2023. Front Cardiovasc Med. 2024. PMID: 38259319 Free PMC article.
-
Short-term transcriptomic response to plasma membrane injury.Sci Rep. 2021 Sep 27;11(1):19141. doi: 10.1038/s41598-021-98420-y. Sci Rep. 2021. PMID: 34580330 Free PMC article.
-
Impact of mechanical cues on key cell functions and cell-nanoparticle interactions.Discov Nano. 2024 Jun 22;19(1):106. doi: 10.1186/s11671-024-04052-2. Discov Nano. 2024. PMID: 38907808 Free PMC article. Review.
References
-
- Lorell, B. H. & Carabello, B. a. Clinical Cardiology: New Frontiers Left Ventricular Hypertrophy. 470–479, 10.1161/01.CIR.102.4.470 (2000). - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

