β-catenin knockdown promotes NHERF1-mediated survival of colorectal cancer cells: implications for a double-targeted therapy
- PMID: 29551770
- PMCID: PMC6002344
- DOI: 10.1038/s41388-018-0170-y
β-catenin knockdown promotes NHERF1-mediated survival of colorectal cancer cells: implications for a double-targeted therapy
Abstract
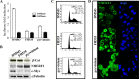
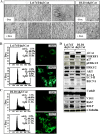
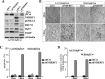
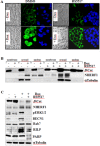
Nuclear activated β-catenin plays a causative role in colorectal cancers (CRC) but remains an elusive therapeutic target. Using human CRC cells harboring different Wnt/β-catenin pathway mutations in APC/KRAS or β-catenin/KRAS genes, and both genetic and pharmacological knockdown approaches, we show that oncogenic β-catenin signaling negatively regulates the expression of NHERF1 (Na+/H+ exchanger 3 regulating factor 1), a PDZ-adaptor protein that is usually lost or downregulated in early dysplastic adenomas to exacerbate nuclear β-catenin activity. Chromatin immunoprecipitation (ChIP) assays demonstrated that β-catenin represses NHERF1 via TCF4 directly, while the association between TCF1 and the Nherf1 promoter increased upon β-catenin knockdown. To note, the occurrence of a cytostatic survival response in settings of single β-catenin-depleted CRC cells was abrogated by combining NHERF1 inhibition via small hairpin RNA (shRNA) or RS5517, a novel PDZ1-domain ligand of NHERF1 that prevented its ectopic nuclear entry. Mechanistically, dual NHERF1/β-catenin targeting promoted an autophagy-to-apoptosis switch consistent with the activation of Caspase-3, the cleavage of PARP and reduced levels of phospho-ERK1/2, Beclin-1, and Rab7 autophagic proteins compared with β-catenin knockdown alone. Collectively, our data unveil novel β-catenin/TCF-dependent mechanisms of CRC carcinogenesis, also offering preclinical proof of concept for combining β-catenin and NHERF1 pharmacological inhibitors as a mechanism-based strategy to augment apoptotic death of CRC cells refractory to current Wnt/β-catenin-targeted therapeutics.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures





References
-
- Frattini M, Balestra D, Suardi S, Oggionni M, Alberici P, Radice P, et al. Different genetic features associated with colon and rectal carcinogenesis. Cancer Res. 2004;10:4015–21. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

