Immunization with outer membrane vesicles displaying conserved surface polysaccharide antigen elicits broadly antimicrobial antibodies
- PMID: 29555731
- PMCID: PMC5889644
- DOI: 10.1073/pnas.1718341115
Immunization with outer membrane vesicles displaying conserved surface polysaccharide antigen elicits broadly antimicrobial antibodies
Abstract
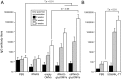
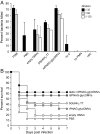
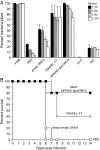
Many microbial pathogens produce a β-(1→6)-linked poly-N-acetyl-d-glucosamine (PNAG) surface capsule, including bacterial, fungal, and protozoan cells. Broadly protective immune responses to this single conserved polysaccharide antigen in animals are possible but only when a deacetylated poly-N-acetyl-d-glucosamine (dPNAG; <30% acetate) glycoform is administered as a conjugate to a carrier protein. Unfortunately, conventional methods for natural extraction or chemical synthesis of dPNAG and its subsequent conjugation to protein carriers can be technically demanding and expensive. Here, we describe an alternative strategy for creating broadly protective vaccine candidates that involved coordinating recombinant poly-N-acetyl-d-glucosamine (rPNAG) biosynthesis with outer membrane vesicle (OMV) formation in laboratory strains of Escherichia coli The glycosylated outer membrane vesicles (glycOMVs) released by these engineered bacteria were decorated with the PNAG glycopolymer and induced high titers of PNAG-specific IgG antibodies after immunization in mice. When a Staphylococcus aureus enzyme responsible for PNAG deacetylation was additionally expressed in these cells, glycOMVs were generated that elicited antibodies to both highly acetylated PNAG (∼95-100% acetate) and a chemically deacetylated dPNAG derivative (∼15% acetate). These antibodies mediated efficient in vitro killing of two distinct PNAG-positive bacterial species, namely S. aureus and Francisella tularensis subsp. holarctica, and mice immunized with PNAG-containing glycOMVs developed protective immunity against these unrelated pathogens. Collectively, our results reveal the potential of glycOMVs for targeting this conserved polysaccharide antigen and engendering protective immunity against the broad range of pathogens that produce surface PNAG.
Keywords: glycoconjugate vaccine; immunization; infectious disease; oligosaccharide; synthetic biology.
Conflict of interest statement
Conflict of interest statement: C.C.-B. is an inventor of intellectual properties (use of human mAb to PNAG and use of PNAG vaccines) that are licensed by Brigham and Women’s Hospital to Alopexx Vaccine, LLC, and Alopexx Pharmaceuticals, LLC. As an inventor of intellectual properties, C.C.-B. also has the right to receive a share of licensing-related income (royalties, fees) through Brigham and Women’s Hospital from Alopexx Pharmaceuticals, LLC, and Alopexx Vaccine, LLC. D.P. and M.P.D. have a financial interest in Versatope, Inc., and M.P.D. also has a financial interest in Glycobia, Inc. The interests of D.P. and M.P.D. are reviewed and managed by Cornell University in accordance with their conflict of interest policies. G.B.P. is an inventor of intellectual properties (human mAb to PNAG and PNAG vaccines) that are licensed by Brigham and Women’s Hospital to Alopexx Vaccine, LLC, and Alopexx Pharmaceuticals, LLC, entities, in which G.B.P. also holds equity. As an inventor of intellectual properties, G.B.P. also has the right to receive a share of licensing-related income (royalties, fees) through Brigham and Women’s Hospital from Alopexx Pharmaceuticals, LLC, and Alopexx Vaccine, LLC. The interests of G.B.P. are reviewed and managed by Brigham and Women’s Hospital and Partners Healthcare in accordance with their conflict of interest policies.
Figures


Similar articles
-
Synthetic {beta}-(1->6)-linked N-acetylated and nonacetylated oligoglucosamines used to produce conjugate vaccines for bacterial pathogens.Infect Immun. 2010 Feb;78(2):764-72. doi: 10.1128/IAI.01093-09. Epub 2009 Nov 30. Infect Immun. 2010. PMID: 19948836 Free PMC article.
-
Outer membrane vesicles displaying engineered glycotopes elicit protective antibodies.Proc Natl Acad Sci U S A. 2016 Jun 28;113(26):E3609-18. doi: 10.1073/pnas.1518311113. Epub 2016 Jun 6. Proc Natl Acad Sci U S A. 2016. PMID: 27274048 Free PMC article.
-
Comparative opsonic and protective activities of Staphylococcus aureus conjugate vaccines containing native or deacetylated Staphylococcal Poly-N-acetyl-beta-(1-6)-glucosamine.Infect Immun. 2005 Oct;73(10):6752-62. doi: 10.1128/IAI.73.10.6752-6762.2005. Infect Immun. 2005. PMID: 16177353 Free PMC article.
-
The exceptionally broad-based potential of active and passive vaccination targeting the conserved microbial surface polysaccharide PNAG.Expert Rev Vaccines. 2016 Aug;15(8):1041-53. doi: 10.1586/14760584.2016.1159135. Epub 2016 Mar 16. Expert Rev Vaccines. 2016. PMID: 26918288 Free PMC article. Review.
-
Broadly protective semi-synthetic glycoconjugate vaccine against pathogens capable of producing poly-β-(1→6)-N-acetyl-d-glucosamine exopolysaccharide.Drug Discov Today Technol. 2020 Dec;35-36:13-21. doi: 10.1016/j.ddtec.2020.09.002. Epub 2020 Oct 26. Drug Discov Today Technol. 2020. PMID: 33388124 Review.
Cited by
-
Marine Delivery Vehicles: Molecular Components and Applications of Bacterial Extracellular Vesicles.Mar Drugs. 2024 Aug 9;22(8):363. doi: 10.3390/md22080363. Mar Drugs. 2024. PMID: 39195479 Free PMC article. Review.
-
Interactions between extracellular vesicles and microbiome in human diseases: New therapeutic opportunities.Imeta. 2023 Feb 6;2(2):e86. doi: 10.1002/imt2.86. eCollection 2023 May. Imeta. 2023. PMID: 38868436 Free PMC article. Review.
-
Recent Progress in Developing Extracellular Vesicles as Nanovehicles to Deliver Carbohydrate-Based Therapeutics and Vaccines.Vaccines (Basel). 2025 Mar 7;13(3):285. doi: 10.3390/vaccines13030285. Vaccines (Basel). 2025. PMID: 40266147 Free PMC article. Review.
-
Outer membrane vesicles as a platform for the discovery of antibodies to bacterial pathogens.Appl Microbiol Biotechnol. 2024 Feb 24;108(1):232. doi: 10.1007/s00253-024-13033-5. Appl Microbiol Biotechnol. 2024. PMID: 38396192 Free PMC article. Review.
-
Pathogenesis Mediated by Bacterial Membrane Vesicles.Subcell Biochem. 2021;97:101-150. doi: 10.1007/978-3-030-67171-6_6. Subcell Biochem. 2021. PMID: 33779916
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

