Preferential Ty1 retromobility in mother cells and nonquiescent stationary phase cells is associated with increased concentrations of total Gag or processed Gag and is inhibited by exposure to a high concentration of calcium
- PMID: 29562219
- PMCID: PMC5892695
- DOI: 10.18632/aging.101402
Preferential Ty1 retromobility in mother cells and nonquiescent stationary phase cells is associated with increased concentrations of total Gag or processed Gag and is inhibited by exposure to a high concentration of calcium
Abstract
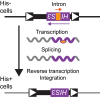
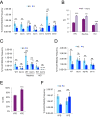
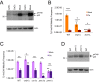
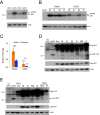
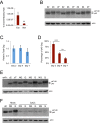
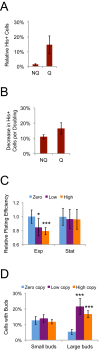
Retrotransposons are abundant mobile DNA elements in eukaryotic genomes that are more active with age in diverse species. Details of the regulation and consequences of retrotransposon activity during aging remain to be determined. Ty1 retromobility in Saccharomyces cerevisiae is more frequent in mother cells compared to daughter cells, and we found that Ty1 was more mobile in nonquiescent compared to quiescent subpopulations of stationary phase cells. This retromobility asymmetry was absent in mutant strains lacking BRP1 that have reduced expression of the essential Pma1p plasma membrane proton pump, lacking the mRNA decay gene LSM1, and in cells exposed to a high concentration of calcium. Mother cells had higher levels of Ty1 Gag protein than daughters. The proportion of protease-processed Gag decreased as cells transitioned to stationary phase, processed Gag was the dominant form in nonquiescent cells, but was virtually absent from quiescent cells. Treatment with calcium reduced total Gag levels and the proportion of processed Gag, particularly in mother cells. We also found that Ty1 reduced the fitness of proliferating but not stationary phase cells. These findings may be relevant to understanding regulation and consequences of retrotransposons during aging in other organisms, due to conserved impacts and regulation of retrotransposons.
Keywords: Saccharomyces cerevisiae; Ty1; quiescence; replicative aging; retrotransposon.
Conflict of interest statement
Figures
Similar articles
-
Preferential retrotransposition in aging yeast mother cells is correlated with increased genome instability.DNA Repair (Amst). 2015 Oct;34:18-27. doi: 10.1016/j.dnarep.2015.07.004. Epub 2015 Aug 7. DNA Repair (Amst). 2015. PMID: 26298836 Free PMC article.
-
The Mediator co-activator complex regulates Ty1 retromobility by controlling the balance between Ty1i and Ty1 promoters.PLoS Genet. 2018 Feb 20;14(2):e1007232. doi: 10.1371/journal.pgen.1007232. eCollection 2018 Feb. PLoS Genet. 2018. PMID: 29462141 Free PMC article.
-
Possible regulatory function of the Saccharomyces cerevisiae Ty1 retrotransposon core protein.Yeast. 2000 Jul;16(10):921-32. doi: 10.1002/1097-0061(200007)16:10<921::AID-YEA588>3.0.CO;2-#. Yeast. 2000. PMID: 10870103
-
Determinants of Genomic RNA Encapsidation in the Saccharomyces cerevisiae Long Terminal Repeat Retrotransposons Ty1 and Ty3.Viruses. 2016 Jul 14;8(7):193. doi: 10.3390/v8070193. Viruses. 2016. PMID: 27428991 Free PMC article. Review.
-
The Ty1 LTR-Retrotransposon of Budding Yeast, Saccharomyces cerevisiae.Microbiol Spectr. 2015 Apr;3(2):MDNA3-0053-2014. doi: 10.1128/microbiolspec.MDNA3-0053-2014. Microbiol Spectr. 2015. PMID: 26104690 Review.
Cited by
-
Diverse transposable element landscapes in pathogenic and nonpathogenic yeast models: the value of a comparative perspective.Mob DNA. 2020 Apr 21;11:16. doi: 10.1186/s13100-020-00215-x. eCollection 2020. Mob DNA. 2020. PMID: 32336995 Free PMC article. Review.
-
A Systematic Review on Quiescent State Research Approaches in S. cerevisiae.Cells. 2023 Jun 12;12(12):1608. doi: 10.3390/cells12121608. Cells. 2023. PMID: 37371078 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

