Wharton's Jelly Mesenchymal Stromal Cells Support the Expansion of Cord Blood-derived CD34+ Cells Mimicking a Hematopoietic Niche in a Direct Cell-cell Contact Culture System
- PMID: 29562783
- PMCID: PMC6434478
- DOI: 10.1177/0963689717737089
Wharton's Jelly Mesenchymal Stromal Cells Support the Expansion of Cord Blood-derived CD34+ Cells Mimicking a Hematopoietic Niche in a Direct Cell-cell Contact Culture System
Abstract
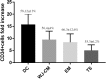
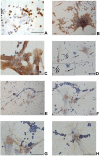
Wharton's jelly mesenchymal stromal cells (WJ-MSCs) have been recently exploited as a feeder layer in coculture systems to expand umbilical cord blood-hematopoietic stem/progenitor cells (UCB-HSPCs). Here, we investigated the role of WJ-MSCs in supporting ex vivo UCB-HSPC expansion either when cultured in direct contact (DC) with WJ-MSCs or separated by a transwell system or in the presence of WJ-MSC-conditioned medium. We found, in short-term culture, a greater degree of expansion of UCB-CD34+ cells in a DC system (15.7 ± 4.1-fold increase) with respect to the other conditions. Moreover, in DC, we evidenced two different CD34+ cell populations (one floating and one adherent to WJ-MSCs) with different phenotypic and functional characteristics. Both multipotent CD34+/CD38- and lineage-committed CD34+/CD38+ hematopoietic progenitors were expanded in a DC system. The former were significantly more represented in the adherent cell fraction than in the floating one (18.7 ± 11.2% vs. 9.7 ± 7.9% over the total CD34+ cells). Short-term colony forming unit (CFU) assays showed that HSPCs adherent to the stromal layer were able to generate a higher frequency of immature colonies (CFU-granulocyte/macrophage and burst-forming unit erythroid/large colonies) with respect to the floating cells. In the attempt to identify molecules that may play a role in supporting the observed ex vivo HSPC growth, we performed secretome analyses. We found a number of proteins involved in the HSPC homing, self-renewal, and differentiation in all tested conditions. It is important to note that a set of sixteen proteins, which are only in part reported to be expressed in any hematopoietic niche, were exclusively found in the DC system secretome. In conclusion, WJ-MSCs allowed a significant ex vivo expansion of multipotent as well as committed HSPCs. This may be relevant for future clinical applications.
Keywords: Wharton’s jelly mesenchymal stromal cells; extracellular matrix; hematopoietic niche; hematopoietic stem and progenitor cell expansion; secretome.
Conflict of interest statement
Figures
Similar articles
-
Wharton's jelly mesenchymal stem cell-based or umbilical vein endothelial cell-based serum-free coculture with cytokines supports the ex vivo expansion/maintenance of cord blood hematopoietic stem/progenitor cells.Stem Cell Res Ther. 2019 Dec 5;10(1):376. doi: 10.1186/s13287-019-1502-8. Stem Cell Res Ther. 2019. PMID: 31806004 Free PMC article.
-
Hypoxia with Wharton's jelly mesenchymal stem cell coculture maintains stemness of umbilical cord blood-derived CD34+ cells.Stem Cell Res Ther. 2018 Jun 13;9(1):158. doi: 10.1186/s13287-018-0902-5. Stem Cell Res Ther. 2018. PMID: 29895317 Free PMC article.
-
Wharton's Jelly Mesenchymal Stromal Cells as a Feeder Layer for the Ex Vivo Expansion of Hematopoietic Stem and Progenitor Cells: a Review.Stem Cell Rev Rep. 2017 Feb;13(1):35-49. doi: 10.1007/s12015-016-9702-4. Stem Cell Rev Rep. 2017. PMID: 27853939 Review.
-
Cord blood CD34+ cells expanded on Wharton's jelly multipotent mesenchymal stromal cells improve the hematopoietic engraftment in NOD/SCID mice.Eur J Haematol. 2014 Nov;93(5):384-91. doi: 10.1111/ejh.12363. Epub 2014 May 26. Eur J Haematol. 2014. PMID: 24797266
-
Characteristics and clinical applications of Wharton's jelly-derived mesenchymal stromal cells.Curr Res Transl Med. 2020 Jan;68(1):5-16. doi: 10.1016/j.retram.2019.09.001. Epub 2019 Sep 19. Curr Res Transl Med. 2020. PMID: 31543433 Review.
Cited by
-
Wharton's jelly mesenchymal stem cells: a concise review of their secretome and prospective clinical applications.Front Cell Dev Biol. 2023 Jun 27;11:1211217. doi: 10.3389/fcell.2023.1211217. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37440921 Free PMC article. Review.
-
Magnetic Nanoparticles and Magnetic Field Exposure Enhances Chondrogenesis of Human Adipose Derived Mesenchymal Stem Cells But Not of Wharton Jelly Mesenchymal Stem Cells.Front Bioeng Biotechnol. 2021 Oct 18;9:737132. doi: 10.3389/fbioe.2021.737132. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34733830 Free PMC article.
-
Human amniotic mesenchymal stromal cells support the ex vivo expansion of cord blood hematopoietic stem cells.Stem Cells Transl Med. 2021 Nov;10(11):1516-1529. doi: 10.1002/sctm.21-0130. Epub 2021 Jul 29. Stem Cells Transl Med. 2021. PMID: 34327849 Free PMC article.
-
An optimized procedure for preparation of conditioned medium from Wharton's jelly mesenchymal stromal cells isolated from umbilical cord.Front Mol Biosci. 2023 Oct 2;10:1273814. doi: 10.3389/fmolb.2023.1273814. eCollection 2023. Front Mol Biosci. 2023. PMID: 37854039 Free PMC article.
-
Umbilical Cord Mesenchymal Stromal Cells for Cartilage Regeneration Applications.Stem Cells Int. 2022 Jan 6;2022:2454168. doi: 10.1155/2022/2454168. eCollection 2022. Stem Cells Int. 2022. PMID: 35035489 Free PMC article. Review.
References
-
- Cohen Y, Nagler A. Umbilical cord blood transplantation-how, when and for whom? Blood. 2004;18(3):167–179. - PubMed
-
- Bari S, Seah KK, Poon Z, Cheunn AM, Fan X, Ong SY, Li S, Koh LP, Hwang WY. Expansion and homing of umbilical cord blood hematopoietic stem and progenitor cells for clinical transplantation. Biol Blood Marrow Transplant. 2015;21(6):1008–1019. - PubMed
-
- Wagner W, Weina F, Roderburga C. Adhesion of hematopoietic progenitor cells to human mesenchymal stem cells as a model for cell-to-cell interaction. Exp Hematol. 2007;35(2):314–325. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

