A Guide for Using Mechanical Stimulation to Enhance Tissue-Engineered Articular Cartilage Properties
- PMID: 29562835
- PMCID: PMC6199627
- DOI: 10.1089/ten.TEB.2018.0006
A Guide for Using Mechanical Stimulation to Enhance Tissue-Engineered Articular Cartilage Properties
Abstract
The use of tissue-engineered articular cartilage (TEAC) constructs has the potential to become a powerful treatment option for cartilage lesions resulting from trauma or early stages of pathology. Although fundamental tissue-engineering strategies based on the use of scaffolds, cells, and signals have been developed, techniques that lead to biomimetic AC constructs that can be translated to in vivo use are yet to be fully confirmed. Mechanical stimulation during tissue culture can be an effective strategy to enhance the mechanical, structural, and cellular properties of tissue-engineered constructs toward mimicking those of native AC. This review focuses on the use of mechanical stimulation to attain and enhance the properties of AC constructs needed to translate these implants to the clinic. In vivo, mechanical loading at maximal and supramaximal physiological levels has been shown to be detrimental to AC through the development of degenerative changes. In contrast, multiple studies have revealed that during culture, mechanical stimulation within narrow ranges of magnitude and duration can produce anisotropic, mechanically robust AC constructs with high cellular viability. Significant progress has been made in evaluating a variety of mechanical stimulation techniques on TEAC, either alone or in combination with other stimuli. These advancements include determining and optimizing efficacious loading parameters (e.g., duration and frequency) to yield improvements in construct design criteria, such as collagen II content, compressive stiffness, cell viability, and fiber organization. With the advancement of mechanical stimulation as a potent strategy in AC tissue engineering, a compendium detailing the results achievable by various stimulus regimens would be of great use for researchers in academia and industry. The objective is to list the qualitative and quantitative effects that can be attained when direct compression, hydrostatic pressure, shear, and tensile loading are used to tissue-engineer AC. Our goal is to provide a practical guide to their use and optimization of loading parameters. For each loading condition, we will also present and discuss benefits and limitations of bioreactor configurations that have been used. The intent is for this review to serve as a reference for including mechanical stimulation strategies as part of AC construct culture regimens.
Keywords: articular cartilage; compression; hydrostatic pressure; mechanical stimulation; shear; tension.
Conflict of interest statement
No competing financial interests exist.
Figures
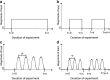
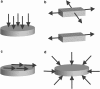

References
-
- Afoke N., Byers P., and Hutton W. Contact pressures in the human hip joint. J Bone Joint Surg Br 69, 536, 1984 - PubMed
-
- Eisenhart R., Adam C., Steinlechner M., and Eckstein F. Quantitative determination of joint incongruity and pressure distribution during simulated gait and cartilage thickness in the human hip joint. J Orthop Res 17, 532, 1999 - PubMed
-
- Li K., Zhang C., Qiu L., Gao L., and Zhang X. Advances in application of mechanical stimuli in bioreactors for cartilage tissue engineering. Tissue Eng Part B Rev 23, 399, 2017 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

