Enzymatically oxidized phospholipids restore thrombin generation in coagulation factor deficiencies
- PMID: 29563336
- PMCID: PMC5926910
- DOI: 10.1172/jci.insight.98459
Enzymatically oxidized phospholipids restore thrombin generation in coagulation factor deficiencies
Abstract
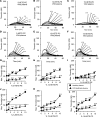
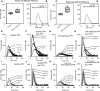
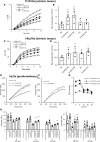
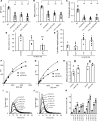
Hemostatic defects are treated using coagulation factors; however, clot formation also requires a procoagulant phospholipid (PL) surface. Here, we show that innate immune cell-derived enzymatically oxidized phospholipids (eoxPL) termed hydroxyeicosatetraenoic acid-phospholipids (HETE-PLs) restore hemostasis in human and murine conditions of pathological bleeding. HETE-PLs abolished blood loss in murine hemophilia A and enhanced coagulation in factor VIII- (FVIII-), FIX-, and FX-deficient human plasma . HETE-PLs were decreased in platelets from patients after cardiopulmonary bypass (CPB). To explore molecular mechanisms, the ability of eoxPL to stimulate individual isolated coagulation factor/cofactor complexes was tested in vitro. Extrinsic tenase (FVIIa/tissue factor [TF]), intrinsic tenase (FVIIIa/FIXa), and prothrombinase (FVa/FXa) all were enhanced by both HETE-PEs and HETE-PCs, suggesting a common mechanism involving the fatty acid moiety. In plasma, 9-, 15-, and 12-HETE-PLs were more effective than 5-, 11-, or 8-HETE-PLs, indicating positional isomer specificity. Coagulation was enhanced at lower lipid/factor ratios, consistent with a more concentrated area for protein binding. Surface plasmon resonance confirmed binding of FII and FX to HETE-PEs. HETE-PEs increased membrane curvature and thickness, but not surface charge or homogeneity, possibly suggesting increased accessibility to cations/factors. In summary, innate immune-derived eoxPL enhance calcium-dependent coagulation factor function, and their potential utility in bleeding disorders is proposed.
Keywords: Coagulation; Hematology.
Conflict of interest statement
Figures


References
-
- Mann KG. Biochemistry and physiology of blood coagulation. Thromb Haemost. 1999;82(2):165–174. - PubMed
-
- Zwaal RF, Schroit AJ. Pathophysiologic implications of membrane phospholipid asymmetry in blood cells. Blood. 1997;89(4):1121–1132. - PubMed
-
- Taylor FB, et al. Lethal E. coli septic shock is prevented by blocking tissue factor with monoclonal antibody. Circ Shock. 1991;33(3):127–134. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

