MFSD2B is a sphingosine 1-phosphate transporter in erythroid cells
- PMID: 29563527
- PMCID: PMC5862976
- DOI: 10.1038/s41598-018-23300-x
MFSD2B is a sphingosine 1-phosphate transporter in erythroid cells
Abstract
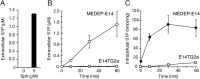
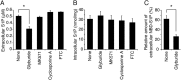
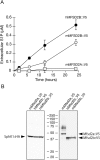
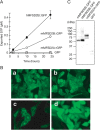
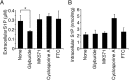
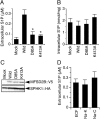
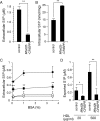
Sphingosine 1-phosphate (S1P) is an intercellular signaling molecule present in blood. Erythrocytes have a central role in maintaining the S1P concentration in the blood stream. We previously demonstrated that S1P is exported from erythrocytes by a glyburide-sensitive S1P transporter. However, the gene encoding the S1P transporter in erythrocytes is unknown. In this study, we found that the mouse erythroid cell line, MEDEP-E14, has S1P export activity and exhibits properties that are consistent with those of erythrocytes. Using microarray analysis of MEDEP-E14 cells and its parental cell line, E14TG2a, we identified several candidate genes for S1P export activity. Of those genes, only one gene, Mfsd2b, showed S1P transport activity. The properties of S1P release by MFSD2B were similar to those in erythrocytes. Moreover, knockout of MFSD2B in MEDEP-E14 cells decreased S1P export from the cells. These results strongly suggest that MFSD2B is a novel S1P transporter in erythroid cells.
Conflict of interest statement
The authors declare no competing interests.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

