Difluoromethylation of (hetero)aryl chlorides with chlorodifluoromethane catalyzed by nickel
- PMID: 29563528
- PMCID: PMC5862906
- DOI: 10.1038/s41467-018-03532-1
Difluoromethylation of (hetero)aryl chlorides with chlorodifluoromethane catalyzed by nickel
Abstract
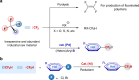
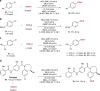
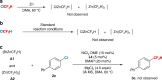
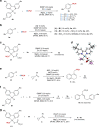
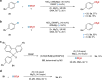
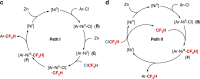
Relatively low reactivity hinders using chlorodifluoromethane (ClCF2H) for general difluoromethylation with organic molecules, despite its availability as an inexpensive industrial chemical. To date, transformations of ClCF2H are very limited and most of them involve difluorocarbene intermediate. Here, we describe a strategy for difluoromethylation of aromatics through nickel-catalyzed cross-coupling of ClCF2H with readily accessible (hetero)aryl chlorides. The reaction proceeds under mild reaction conditions with high efficiency and features synthetic simplicity without preformation of arylmetals and broad substrate scope, including a variety of heteroaromatics and commercially available pharmaceuticals. The reliable practicability and scalability of the current nickel-catalyzed process has also been demonstrated by several 10-g scale reactions without loss of reaction efficiency. Preliminary mechanistic studies reveal that the reaction starts from the oxidative addition of aryl chlorides to Ni(0) and a difluoromethyl radical is involved in the reaction, providing a route for applications of ClCF2H in organic synthesis and related chemistry.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Hudlicky, M. & Pavlath, A. E. Chemistry of Organic Fluorine Compounds II. (American Chemical Society, Washington, DC, 1995).
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

