Optimizing labelling conditions of 213Bi-DOTATATE for preclinical applications of peptide receptor targeted alpha therapy
- PMID: 29564386
- PMCID: PMC5843812
- DOI: 10.1186/s41181-016-0014-4
Optimizing labelling conditions of 213Bi-DOTATATE for preclinical applications of peptide receptor targeted alpha therapy
Erratum in
-
Erratum: Publisher Correction to EJNMMI Radiopharmacy and Chemistry Volume 1 (2016).EJNMMI Radiopharm Chem. 2018 Nov 26;3:13. doi: 10.1186/s41181-018-0049-9. eCollection 2018 Dec. EJNMMI Radiopharm Chem. 2018. PMID: 31329807 Free PMC article.
Abstract
Background: 213Bismuth (213Bi, T1/2 = 45.6 min) is one of the most frequently used α-emitters in cancer research. High specific activity radioligands are required for peptide receptor radionuclide therapy. The use of generators containing less than 222 MBq 225Ac (actinium), due to limited availability and the high cost to produce large-scale 225Ac/213Bi generators, might complicate in vitro and in vivo applications though.Here we present optimized labelling conditions of a DOTA-peptide with an 225Ac/213Bi generator (< 222 MBq) for preclinical applications using DOTA-Tyr3-octreotate (DOTATATE), a somatostatin analogue. The following labelling conditions of DOTATATE with 213Bi were investigated; peptide mass was varied from 1.7 to 7.0 nmol, concentration of TRIS buffer from 0.15 mol.L-1 to 0.34 mol.L-1, and ascorbic acid from 0 to 71 mmol.L-1 in 800 μL. All reactions were performed at 95 °C for 5 min. After incubation, DTPA (50 nmol) was added to stop the labelling reaction. Besides optimizing the labelling conditions, incorporation yield was determined by ITLC-SG and radiochemical purity (RCP) was monitored by RP-HPLC up to 120 min after labelling. Dosimetry studies in the reaction vial were performed using Monte Carlo and in vitro clonogenic assay was performed with a rat pancreatic tumour cell line, CA20948.
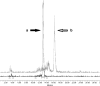
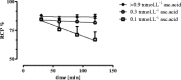
Results: At least 3.5 nmol DOTATATE was required to obtain incorporation ≥ 99 % with 100 MBq 213Bi (at optimized pH conditions, pH 8.3 with 0.15 mol.L-1 TRIS) in a reaction volume of 800 μL. The cumulative absorbed dose in the reaction vial was 230 Gy/100 MBq in 30 min. A minimal final concentration of 0.9 mmol.L-1 ascorbic acid was required for ~100 MBq (t = 0) to minimize radiation damage of DOTATATE. The osmolarity was decreased to 0.45 Osmol/L.Under optimized labelling conditions, 213Bi-DOTATATE remained stable up to 2 h after labelling, RCP was ≥ 85 %. In vitro showed a negative correlation between ascorbic acid concentration and cell survival.
Conclusion: 213Bismuth-DOTA-peptide labelling conditions including peptide amount, quencher and pH were optimized to meet the requirements needed for preclinical applications in peptide receptor radionuclide therapy.
Keywords: 213Bismuth; Absorbed dose; Ascorbic acid; Radiochemical purity; Somatostatin; Targeted alpha therapy.
Figures




References
-
- Bibler NE. Gamma and alpha radiolysis of aqueous solutions of diethylenetriaminepenta acetic acid. J Inorg Nucl Chem. 1972;34:8. doi: 10.1016/0022-1902(72)80342-7. - DOI
-
- Brechbiel MW. Chelated metal ions for therapeutic and diagnostic applications. Exp Biol Med (Maywood) 2001;226(7):627–628. - PubMed
-
- Breeman WA, Kwekkeboom DJ, Kooij PP, Bakker WH, Hofland LJ, Visser TJ, et al. Effect of dose and specific activity on tissue distribution of indium-111-pentetreotide in rats. J Nucl Med. 1995;36(4):623–627. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
