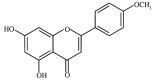
Licochalcone D induces apoptosis and inhibits migration and invasion in human melanoma A375 cells
- PMID: 29565458
- PMCID: PMC5928765
- DOI: 10.3892/or.2018.6329
Licochalcone D induces apoptosis and inhibits migration and invasion in human melanoma A375 cells
Abstract
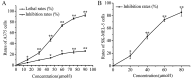
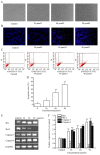
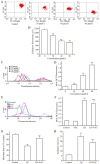
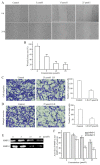
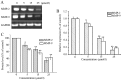
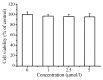
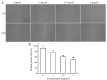
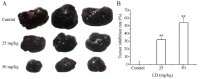
The aim of the present study was to determine the effects of Licochalcone D (LD) on the apoptosis and migration and invasion in human melanoma A375 cells. Cell proliferation was determined by sulforhodamine B assay. Apoptosis was assessed by Hoechst 33258 and Annexin V‑FITC/PI staining and JC‑1 assay. Total intracellular reactive oxygen species (ROS) was examined by DCFH‑DA. Wound healing and Transwell assays were used to detect migration and invasion of the cells. The activities of matrix metalloproteinase (MMP‑2 and MMP‑9) were assessed via gelatin zymography. Tumor growth in vivo was evaluated in C57BL/6 mice. RT‑PCR, qPCR, ELISA and western blot analysis were utilized to measure the mRNA and protein levels. Our results showed that LD inhibited the proliferation of A375 and SK‑MEL‑5 cells in a concentration‑dependent manner. After treatment with LD, A375 cells displayed obvious apoptotic characteristics, and the number of apoptotic cells was significantly increased. Pro‑apoptotic protein Bax, caspase‑9 and caspase‑3 were upregulated, while anti‑apoptotic protein Bcl‑2 was downregulated in the LD‑treated cells. Meanwhile, LD induced the loss of mitochondrial membrane potential (ΔΨm) and increased the level of ROS. ROS production was inhibited by the co‑treatment of LD and free radical scavenger N‑acetyl‑cysteine (NAC). Furthermore, LD also blocked A375 cell migration and invasion in vitro which was associated with the downregulation of MMP‑9 and MMP‑2. Finally, intragastric administration of LD suppressed tumor growth in the mouse xenograft model of murine melanoma B16F0 cells. These results suggest that LD may be a potential drug for human melanoma treatment by inhibiting proliferation, inducing apoptosis via the mitochondrial pathway and blocking cell migration and invasion.
Figures
Similar articles
-
Luteolin inhibits proliferation and induces apoptosis of human melanoma cells in vivo and in vitro by suppressing MMP-2 and MMP-9 through the PI3K/AKT pathway.Food Funct. 2019 Feb 20;10(2):703-712. doi: 10.1039/c8fo02013b. Food Funct. 2019. PMID: 30663726
-
Casticin Inhibits A375.S2 Human Melanoma Cell Migration/Invasion through Downregulating NF-κB and Matrix Metalloproteinase-2 and -1.Molecules. 2016 Mar 19;21(3):384. doi: 10.3390/molecules21030384. Molecules. 2016. PMID: 27007357 Free PMC article.
-
Hinokiflavone induces apoptosis in melanoma cells through the ROS-mitochondrial apoptotic pathway and impairs cell migration and invasion.Biomed Pharmacother. 2018 Jul;103:101-110. doi: 10.1016/j.biopha.2018.02.076. Epub 2018 Apr 7. Biomed Pharmacother. 2018. PMID: 29635122
-
Cryptotanshinone induces melanoma cancer cells apoptosis via ROS-mitochondrial apoptotic pathway and impairs cell migration and invasion.Biomed Pharmacother. 2016 Aug;82:319-26. doi: 10.1016/j.biopha.2016.05.015. Epub 2016 May 24. Biomed Pharmacother. 2016. PMID: 27470369
-
Therapeutic Potential of Silver Nitroprusside Nanoparticles for Melanoma.ACS Appl Bio Mater. 2024 Aug 19;7(8):5057-5075. doi: 10.1021/acsabm.4c00597. Epub 2024 Aug 8. ACS Appl Bio Mater. 2024. PMID: 39115261 Review.
Cited by
-
Chalcone-Acridine Hybrid Suppresses Melanoma Cell Progression via G2/M Cell Cycle Arrest, DNA Damage, Apoptosis, and Modulation of MAP Kinases Activity.Int J Mol Sci. 2022 Oct 14;23(20):12266. doi: 10.3390/ijms232012266. Int J Mol Sci. 2022. PMID: 36293123 Free PMC article.
-
Anticancer effects of licochalcones: A review of the mechanisms.Front Pharmacol. 2023 Jan 23;14:1074506. doi: 10.3389/fphar.2023.1074506. eCollection 2023. Front Pharmacol. 2023. PMID: 36755942 Free PMC article.
-
Glycyrrhiza glabra (Licorice): A Comprehensive Review on Its Phytochemistry, Biological Activities, Clinical Evidence and Toxicology.Plants (Basel). 2021 Dec 14;10(12):2751. doi: 10.3390/plants10122751. Plants (Basel). 2021. PMID: 34961221 Free PMC article. Review.
-
Exploration potential sepsis-ferroptosis mechanisms through the use of CETSA technology and network pharmacology.Sci Rep. 2025 Apr 19;15(1):13527. doi: 10.1038/s41598-025-95451-7. Sci Rep. 2025. PMID: 40253433 Free PMC article.
-
Licochalcone D mitigates intracerebral hemorrhage-induced ferroptosis of neurons through COX2 inhibition.Front Pharmacol. 2025 Jul 8;16:1566724. doi: 10.3389/fphar.2025.1566724. eCollection 2025. Front Pharmacol. 2025. PMID: 40697659 Free PMC article.
References
-
- Reuland SN, Goldstein NB, Partyka KA, Cooper DA, Fujita M, Norris DA, Shellman YG. The combination of BH3-mimetic ABT-737 with the alkylating agent temozolomide induces strong synergistic killing of melanoma cells independent of p53. PLoS One. 2011;6:e24294. doi: 10.1371/journal.pone.0024294. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

