Use of chitin and chitosan to produce new chitooligosaccharides by chitinase Chit42: enzymatic activity and structural basis of protein specificity
- PMID: 29566690
- PMCID: PMC5863366
- DOI: 10.1186/s12934-018-0895-x
Use of chitin and chitosan to produce new chitooligosaccharides by chitinase Chit42: enzymatic activity and structural basis of protein specificity
Abstract
Background: Chitinases are ubiquitous enzymes that have gained a recent biotechnological attention due to their ability to transform biological waste from chitin into valued chito-oligomers with wide agricultural, industrial or medical applications. The biological activity of these molecules is related to their size and acetylation degree. Chitinase Chit42 from Trichoderma harzianum hydrolyses chitin oligomers with a minimal of three N-acetyl-D-glucosamine (GlcNAc) units. Gene chit42 was previously characterized, and according to its sequence, the encoded protein included in the structural Glycoside Hydrolase family GH18.
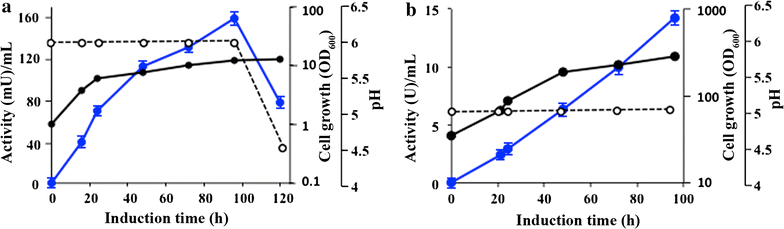
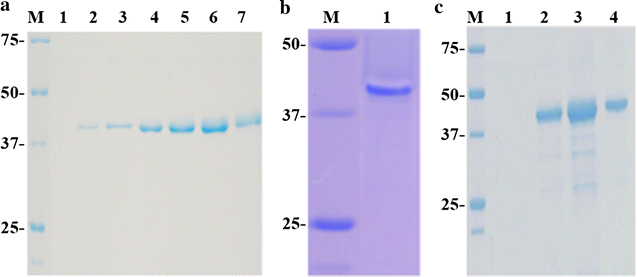
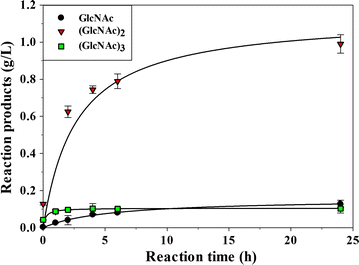
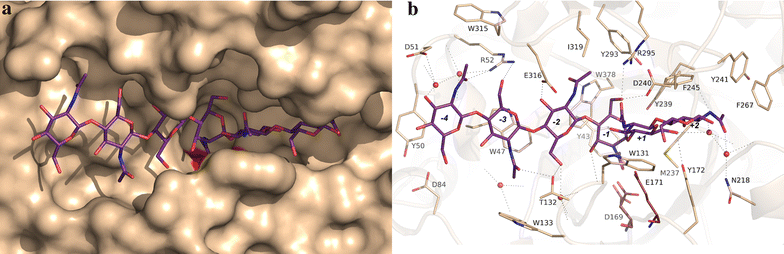
Results: Chit42 was expressed in Pichia pastoris using fed-batch fermentation to about 3 g/L. Protein heterologously expressed showed similar biochemical properties to those expressed by the natural producer (42 kDa, optima pH 5.5-6.5 and 30-40 °C). In addition to hydrolyse colloidal chitin, this enzyme released reducing sugars from commercial chitosan of different sizes and acetylation degrees. Chit42 hydrolysed colloidal chitin at least 10-times more efficiently (defined by the kcat/Km ratio) than any of the assayed chitosan. Production of partially acetylated chitooligosaccharides was confirmed in reaction mixtures using HPAEC-PAD chromatography and mass spectrometry. Masses corresponding to (D-glucosamine)1-8-GlcNAc were identified from the hydrolysis of different substrates. Crystals from Chit42 were grown and the 3D structure determined at 1.8 Å resolution, showing the expected folding described for other GH18 chitinases, and a characteristic groove shaped substrate-binding site, able to accommodate at least six sugar units. Detailed structural analysis allows depicting the features of the Chit42 specificity, and explains the chemical nature of the partially acetylated molecules obtained from analysed substrates.
Conclusions: Chitinase Chit42 was expressed in a heterologous system to levels never before achieved. The enzyme produced small partially acetylated chitooligosaccharides, which have enormous biotechnological potential in medicine and food. Chit42 3D structure was characterized and analysed. Production and understanding of how the enzymes generating bioactive chito-oligomers work is essential for their biotechnological application, and paves the way for future work to take advantage of chitinolytic activities.
Keywords: Chit42 3D structure; Chitinase; Chitooligosaccharides; Partially acetylated chitooligosaccharides; Trichoderma harzianum.
Figures


Similar articles
-
Chitinous material bioconversion by three new chitinases from the yeast Mestchnikowia pulcherrima.Microb Cell Fact. 2024 Jan 20;23(1):31. doi: 10.1186/s12934-024-02300-9. Microb Cell Fact. 2024. PMID: 38245740 Free PMC article. Review.
-
Endo-chitinase Chit33 specificity on different chitinolytic materials allows the production of unexplored chitooligosaccharides with antioxidant activity.Biotechnol Rep (Amst). 2020 Jul 4;27:e00500. doi: 10.1016/j.btre.2020.e00500. eCollection 2020 Sep. Biotechnol Rep (Amst). 2020. PMID: 32685384 Free PMC article.
-
Structural inspection and protein motions modelling of a fungal glycoside hydrolase family 18 chitinase by crystallography depicts a dynamic enzymatic mechanism.Comput Struct Biotechnol J. 2021 Oct 2;19:5466-5478. doi: 10.1016/j.csbj.2021.09.027. eCollection 2021. Comput Struct Biotechnol J. 2021. PMID: 34712392 Free PMC article.
-
Heterologous Expression and Characterization of a pH-Stable Chitinase from Micromonospora aurantiaca with a Potential Application in Chitin Degradation.Mar Drugs. 2024 Jun 20;22(6):287. doi: 10.3390/md22060287. Mar Drugs. 2024. PMID: 38921598 Free PMC article.
-
Conversion of Chitin to Defined Chitosan Oligomers: Current Status and Future Prospects.Mar Drugs. 2019 Aug 1;17(8):452. doi: 10.3390/md17080452. Mar Drugs. 2019. PMID: 31374920 Free PMC article. Review.
Cited by
-
Chitinases: Therapeutic Scaffolds for Allergy and Inflammation.Recent Pat Inflamm Allergy Drug Discov. 2020;14(1):46-57. doi: 10.2174/1872213X14666200114184054. Recent Pat Inflamm Allergy Drug Discov. 2020. PMID: 31934842 Free PMC article. Review.
-
Progress and challenges in the synthesis of sequence controlled polysaccharides.Beilstein J Org Chem. 2021 Aug 5;17:1981-2025. doi: 10.3762/bjoc.17.129. eCollection 2021. Beilstein J Org Chem. 2021. PMID: 34386106 Free PMC article. Review.
-
Seafood Waste as Attractive Source of Chitin and Chitosan Production and Their Applications.Int J Mol Sci. 2020 Jun 16;21(12):4290. doi: 10.3390/ijms21124290. Int J Mol Sci. 2020. PMID: 32560250 Free PMC article. Review.
-
Chitinous material bioconversion by three new chitinases from the yeast Mestchnikowia pulcherrima.Microb Cell Fact. 2024 Jan 20;23(1):31. doi: 10.1186/s12934-024-02300-9. Microb Cell Fact. 2024. PMID: 38245740 Free PMC article. Review.
-
Structure-Function Insights into the Fungal Endo-Chitinase Chit33 Depict its Mechanism on Chitinous Material.Int J Mol Sci. 2022 Jul 9;23(14):7599. doi: 10.3390/ijms23147599. Int J Mol Sci. 2022. PMID: 35886948 Free PMC article.
References
-
- Rinaudo M. Chitin and chitosan: properties and applications. Prog Polym Sci. 2006;31(7):603–632. doi: 10.1016/j.progpolymsci.2006.06.001. - DOI
-
- Khalil IR, Burns AT, Radecka I, Kowalczuk M, Khalaf T, Adamus G, Johnston B, Khechara MP. Bacterial-derived polymer poly-γ-glutamic acid (γ-PGA)-based micro/nanoparticles as a delivery system for antimicrobials and other biomedical applications. Int J Mol Sci. 2017;18(2):1–18. doi: 10.3390/ijms18020313. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

