Ponatinib efficacy and safety in Philadelphia chromosome-positive leukemia: final 5-year results of the phase 2 PACE trial
- PMID: 29567798
- PMCID: PMC6071555
- DOI: 10.1182/blood-2016-09-739086
Ponatinib efficacy and safety in Philadelphia chromosome-positive leukemia: final 5-year results of the phase 2 PACE trial
Abstract
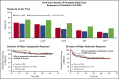
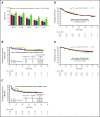
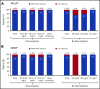
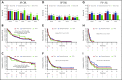
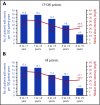
Ponatinib has potent activity against native and mutant BCR-ABL1, including BCR-ABL1T315I The pivotal phase 2 Ponatinib Ph+ ALL and CML Evaluation (PACE) trial evaluated efficacy and safety of ponatinib at a starting dose of 45 mg once daily in 449 patients with chronic myeloid leukemia (CML) or Philadelphia chromosome-positive acute lymphoblastic leukemia (ALL) resistant/intolerant to dasatinib or nilotinib, or with BCR-ABL1T315I This analysis focuses on chronic-phase CML (CP-CML) patients (n = 270) with 56.8-month median follow-up. Among 267 evaluable patients, 60%, 40%, and 24% achieved major cytogenetic response (MCyR), major molecular response (MMR), and 4.5-log molecular response, respectively. The probability of maintaining MCyR for 5 years was 82% among responders. Dose reductions were implemented in October 2013 to decrease the risk of arterial occlusive events (AOEs); ≥90% of CP-CML patients who had achieved MCyR or MMR maintained response 40 months after elective dose reductions. Estimated 5-year overall survival was 73%. In CP-CML patients, the most common treatment-emergent adverse events were rash (47%), abdominal pain (46%), thrombocytopenia (46%), headache (43%), dry skin (42%), and constipation (41%). The cumulative incidence of AOEs in CP-CML patients increased over time to 31%, while the exposure-adjusted incidence of new AOEs (15.8 and 4.9 per 100 patient-years in years 1 and 5, respectively) did not increase over time. These final PACE results demonstrate ponatinib provides durable and clinically meaningful responses, irrespective of dose reductions, in this population of heavily pretreated CP-CML patients. This trial was registered at www.clinicaltrials.gov as #NCT01207440.
© 2018 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: J.E.C. has received research funding from ARIAD, Bristol-Myers Squibb, Novartis, Pfizer, and Teva, and has served as a consultant for ARIAD, Bristol-Myers Squibb, Novartis, and Pfizer. D.-W.K. has received research funding from ARIAD. J.P.-I. has received honoraria from Bristol-Myers Squibb; has received research funding from ARIAD and Novartis; and has served as a consultant or speaker for ARIAD, Bristol-Myers Squibb, Novartis, Pfizer, and Teva. P.D.l.C. has received honoraria from ARIAD, Bristol-Myers Squibb, Novartis, and Pfizer, and research funding from ARIAD. R.P. has received honoraria from, and has served as a speaker for, ARIAD, Bristol-Myers Squibb, Incyte, and Novartis; has served in a consulting or advisory role for AstraZeneca; and has received research funding from ARIAD. C.C. has received research funding from ARIAD. F.E.N. has received honoraria from ARIAD, Bristol-Myers Squibb, and Novartis; has served in a consulting or advisory role for Bristol-Myers Squibb and Novartis; has served on speakers’ bureaus for ARIAD, Bristol-Myers Squibb, and Novartis; has received research funding from ARIAD and Novartis; and has received travel, accommodations, or other expense reimbursements from Bristol-Myers Squibb and Novartis. J.F.A. has received honoraria from ARIAD, Bristol-Myers Squibb, MSD, Novartis, and Pfizer, and has received research funding from ARIAD and Novartis. H.J.K. received honoraria and research funding from ARIAD. M.T. has served in a consulting or advisory role for ARIAD, Novartis, and Pfizer; has received research funding from ARIAD, Incyte, Novartis, Pfizer, and Sanofi; and has received travel, accommodations, or other expense reimbursements from ARIAD, Incyte, Novartis, and Pfizer. D.J.D. has served in a consulting or advisory role for Amgen, ARIAD, Bristol-Myers Squibb, Incyte, Novartis, and Pfizer, and has received research funding from ARIAD. E.A. has served in a consulting or advisory role for ARIAD, Bristol-Myers Squibb, Novartis, and Pfizer, and has received research funding from ARIAD. D.R. has received honoraria for nonpromotional oral presentations from, and has served on advisory boards for, ARIAD, Bristol-Myers Squibb, Novartis, and Pfizer, and has received research funding from ARIAD. M.B. has served as a consultant and speaker for, and received honoraria from, ARIAD, Bristol-Myers Squibb, Novartis, and Pfizer, and has received research funding from ARIAD. M.C.M. has received honoraria for consulting or advisory roles and research funding from ARIAD, Bristol-Myers Squibb, and Novartis. C.G.-P. has served in an advisory role to Bristol-Myers Squibb and Pfizer and has received research funding from ARIAD. S.L. is an employee of, and may own stock/stock options in, ARIAD. V.M.R. is an employee of, and may own stock/stock options in, ARIAD. F.G.H. was an employee of, and may have owned stock/stock options in, ARIAD at the time the work was conducted. F.G. has received honoraria from Novartis and Pfizer; has served in a consulting or advisory role for Celgene; has received travel, accommodations, or other expense reimbursements from Novartis; and has received research funding from ARIAD. M.W.D. has served in a consulting or advisory role for ARIAD, Bristol-Myers Squibb, Incyte, Novartis, and Pfizer; has received travel, accommodations, or other expense reimbursements from ARIAD, Bristol-Myers Squibb, CTI BioPharma, Novartis, and Pfizer; has provided expert testimony for Bristol-Myers Squibb; has received honoraria from ARIAD, Bristol-Myers Squibb, CTI BioPharma, Incyte, Novartis, and Pfizer; and has received research funding from ARIAD, Bristol-Myers Squibb, Celgene, Incyte, and Novartis. A.H. has received research funding from ARIAD, Bristol-Myers Squibb, Novartis, and Pfizer. T.P.H. has received honoraria from, has served in a consulting or advisory role for, and has received research funding from ARIAD, Bristol-Myers Squibb, and Novartis, and has received travel, accommodations, or other expense reimbursements from Bristol-Myers Squibb and Novartis. N.P.S. has received research funding from ARIAD, Bristol-Myers Squibb, Daiichi-Sankyo, Pfizer, and Plexxikon. H.M.K. has received research funding from Amgen, Bristol-Myers Squibb, Novartis, and Pfizer.
Figures
References
-
- Iclusig [package insert]. Cambridge, MA: ARIAD Pharmaceuticals, Inc, a wholly owned subsidiary of Takeda Pharmaceutical Company Limited; 2016.
-
- Iclusig [summary of product characteristics]. Leatherhead, United Kingdom: ARIAD Pharmaceuticals, Inc, a wholly owned subsidiary of Takeda Pharmaceutical Company Limited; 2017.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

