Novel assay demonstrates that coronary artery disease patients have heightened procoagulant platelet response
- PMID: 29569428
- PMCID: PMC6635759
- DOI: 10.1111/jth.14008
Novel assay demonstrates that coronary artery disease patients have heightened procoagulant platelet response
Abstract
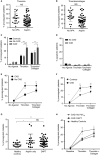
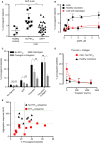
Essentials Procoagulant platelets can be detected using GSAO in human whole blood. Stable coronary artery disease is associated with a heightened procoagulant platelet response. Agonist-induced procoagulant platelet response is not inhibited by aspirin alone. Collagen plus thrombin induced procoagulant platelet response is partially resistant to clopidogrel.
Summary: Background Procoagulant platelets are a subset of highly activated platelets with a critical role in thrombin generation. Evaluation of their clinical utility in thrombotic disorders, such as coronary artery disease (CAD), has been thwarted by the lack of a sensitive and specific whole blood assay. Objectives We developed a novel assay, utilizing the cell death marker, GSAO [(4-(N-(S-glutathionylacetyl)amino)phenylarsonous acid], and the platelet activation marker, P-selectin, to identify procoagulant platelets in whole blood by flow cytometry. Patients/Methods Using this assay, we characterized the procoagulant platelet population in healthy controls and a cohort of patients undergoing elective coronary angiography. Results In patients with CAD, compared with patients without CAD, there was a heightened procoagulant platelet response to thrombin (25.2% vs. 12.2%), adenosine diphosphate (ADP) (7.8% vs. 2.7%) and thrombin plus collagen (27.2% vs. 18.3%). The heightened procoagulant platelet potential in CAD patients was not associated with other markers of platelet function, including aggregation, dense granule release and activation of α2b β3 integrin. Although dual antiplatelet therapy (DAPT) was associated with partial suppression of procoagulant platelets, this inhibitory effect on a patient level could not be predicted by aggregation response to ADP and was not fully suppressed by clopidogrel. Conclusions We report for the first time that procoagulant platelets can be efficiently detected in a few microliters of whole blood using the cell death marker, GSAO, and the platelet activation marker, P-selectin. A heightened procoagulant platelet response may provide insight into the thrombotic risk of CAD and help identify a novel target for antiplatelet therapies in CAD.
Keywords: coronary artery disease; flow cytometry; platelet activation; platelet aggregation inhibitors; platelet function studies; procoagulant platelets.
© 2018 The Authors. Journal of Thrombosis and Haemostasis published by Wiley Periodicals, Inc. on behalf of International Society on Thrombosis and Haemostasis.
Conflict of interest statement
L. Pasalic, V. M. Chen and P. J. Hogg have patent PCT/AU2015/000638 pending. The other authors state that they have no conflict of interest.
Figures


References
-
- Heemskerk JWM, Mattheij NJA, Cosemans JMEM. Platelet‐based coagulation: different populations, different functions. J Thromb Haemost 2013; 11: 2–16. - PubMed
-
- McFadyen JD, Jackson SP. Differentiating haemostasis from thrombosis for therapeutic benefit. Thromb Haemost 2013; 110: 859–67. - PubMed
-
- Bouchard BA, Silveira JR, Tracy PB. Chapter 21 ‐ interactions between platelets and the coagulation system In: Michelson AD, ed. Platelets (3rd Edn). Cambridge: Academic Press, 2013: 425–51.
-
- Munnix ICA, Kuijpers MJE, Auger J, Thomassen CMLGD, Panizzi P, van Zandvoort MAM, Rosing J, Bock PE, Watson SP, Heemskerk JWM. Segregation of platelet aggregatory and procoagulant microdomains in thrombus formation: regulation by transient integrin activation. Arterioscler Thromb Vasc Biol 2007; 27: 2484–90. - PMC - PubMed
-
- Libby P, Theroux P. Pathophysiology of coronary artery disease. Circulation 2005; 111: 3481–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

