Impairment of an Endothelial NAD+-H2S Signaling Network Is a Reversible Cause of Vascular Aging
- PMID: 29570999
- PMCID: PMC5884172
- DOI: 10.1016/j.cell.2018.02.008
Impairment of an Endothelial NAD+-H2S Signaling Network Is a Reversible Cause of Vascular Aging
Erratum in
-
Impairment of an Endothelial NAD+-H2S Signaling Network Is a Reversible Cause of Vascular Aging.Cell. 2019 Feb 7;176(4):944-945. doi: 10.1016/j.cell.2019.01.026. Cell. 2019. PMID: 30735637 Free PMC article. No abstract available.
Abstract
A decline in capillary density and blood flow with age is a major cause of mortality and morbidity. Understanding why this occurs is key to future gains in human health. NAD precursors reverse aspects of aging, in part, by activating sirtuin deacylases (SIRT1-SIRT7) that mediate the benefits of exercise and dietary restriction (DR). We show that SIRT1 in endothelial cells is a key mediator of pro-angiogenic signals secreted from myocytes. Treatment of mice with the NAD+ booster nicotinamide mononucleotide (NMN) improves blood flow and increases endurance in elderly mice by promoting SIRT1-dependent increases in capillary density, an effect augmented by exercise or increasing the levels of hydrogen sulfide (H2S), a DR mimetic and regulator of endothelial NAD+ levels. These findings have implications for improving blood flow to organs and tissues, increasing human performance, and reestablishing a virtuous cycle of mobility in the elderly.
Keywords: NAD(+); aging; angiogenesis; endurance; exercise; hydrogen sulfide; ischemia; nicotinamide mononucleotide; sirtuins; skeletal muscle capillaries.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures

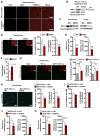
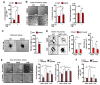

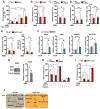
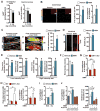
Comment in
-
H2S to Mitigate Vascular Aging: A SIRT1 Connection.Cell. 2018 Mar 22;173(1):8-10. doi: 10.1016/j.cell.2018.03.011. Cell. 2018. PMID: 29571000
References
-
- Arany Z, Foo SY, Ma Y, Ruas JL, Bommi-Reddy A, Girnun G, Cooper M, Laznik D, Chinsomboon J, Rangwala SM, et al. HIF-independent regulation of VEGF and angiogenesis by the transcriptional coactivator PGC-1alpha. Nature. 2008;451:1008–1012. - PubMed
-
- Baker M, Robinson SD, Lechertier T, Barber PR, Tavora B, D’Amico G, Jones DT, Vojnovic B, Hodivala-Dilke K. Use of the mouse aortic ring assay to study angiogenesis. Nat Protoc. 2012;7:89–104. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

