PDK1 governs thromboxane generation and thrombosis in platelets by regulating activation of Raf1 in the MAPK pathway
- PMID: 29575487
- PMCID: PMC5984143
- DOI: 10.1111/jth.14005
PDK1 governs thromboxane generation and thrombosis in platelets by regulating activation of Raf1 in the MAPK pathway
Abstract
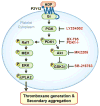
Essentials Phosphoinositide 3-kinase and MAPK pathways crosstalk via PDK1. PDK1 is required for adenosine diphosphate-induced platelet activation and thromboxane generation. PDK1 regulates RAF proto-oncogene Ser/Thr kinase (Raf1) activation in the MAPK pathway. Genetic ablation of PDK1 protects against platelet-dependent thrombosis in vivo.
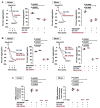
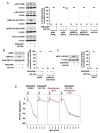
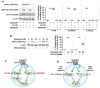
Summary: Background Platelets are dynamic effector cells with functions that span hemostatic, thrombotic and inflammatory continua. Phosphoinositide-dependent protein kinase 1 (PDK1) regulates protease-activated receptor 4-induced platelet activation and thrombus formation through glycogen synthase kinase3β. However, whether PDK1 also signals through the ADP receptor and its functional importance in vivo remain unknown. Objective To establish the mechanism of PDK1 in ADP-induced platelet activation and thrombosis. Methods We assessed the role of PDK1 on 2MeSADP-induced platelet activation by measuring aggregation, thromboxane generation and phosphorylation events in the presence of BX-795, which inhibits PDK1, or by using platelet-specific PDK1 knockout mice and performing western blot analysis. PDK1 function in thrombus formation was assessed with an in vivo pulmonary embolism model. Results PDK1 inhibition with BX-795 reduced 2-methylthio-ADP (2MeSADP)-induced aggregation of human and murine platelets by abolishing thromboxane generation. Similar results were observed in pdk1-/- mice. PDK1 was also necessary for the phosphorylation of mitogen-activated protein kinase kinase 1/2 (MEK1/2), extracellular signal-regulated kinase 1/2, and cytosolic phospholipase A2, indicating that PDK1 regulates an upstream kinase in the mitogen-activated protein kinase (MAPK) pathway. We next determined that this upstream kinase is Raf-1, a serine/threonine kinase that is necessary for the phosphorylation of MEK1/2, as pharmacological inhibition and genetic ablation of PDK1 were sufficient to prevent Raf1 phosphorylation. Furthermore, in vivo inhibition or genetic ablation of PDK1 protected mice from collagen/epinephrine-induced pulmonary embolism. Conclusion PDK1 governs thromboxane generation and thrombosis in platelets that are stimulated with 2MeSADP by regulating activation of the MAPK pathway.
Keywords: phosphoinositide dependent kinase 1; platelet activation; purinergic receptor P2Y12; thrombosis; thromboxane A2.
© 2018 International Society on Thrombosis and Haemostasis.
Conflict of interest statement
The authors state that they have no conflict of interest.
Figures



Comment in
-
PDK1 governs thromboxane generation and thrombosis in platelets by regulating activation of Raf1 in the MAPK pathway: reply.J Thromb Haemost. 2018 Sep;16(9):1904-1905. doi: 10.1111/jth.14230. Epub 2018 Aug 16. J Thromb Haemost. 2018. PMID: 29981265 Free PMC article. No abstract available.
-
PDK1 governs thromboxane generation and thrombosis in platelets by regulating activation of Raf1 in the MAPK pathway: comment.J Thromb Haemost. 2018 Sep;16(9):1901-1904. doi: 10.1111/jth.14232. Epub 2018 Aug 16. J Thromb Haemost. 2018. PMID: 29983001 Free PMC article. No abstract available.
Similar articles
-
The small-molecule MERTK inhibitor UNC2025 decreases platelet activation and prevents thrombosis.J Thromb Haemost. 2018 Feb;16(2):352-363. doi: 10.1111/jth.13875. Epub 2018 Jan 12. J Thromb Haemost. 2018. PMID: 29045015 Free PMC article.
-
PDK1 Determines Collagen-Dependent Platelet Ca2+ Signaling and Is Critical to Development of Ischemic Stroke In Vivo.Arterioscler Thromb Vasc Biol. 2016 Aug;36(8):1507-16. doi: 10.1161/ATVBAHA.115.307105. Epub 2016 Jun 23. Arterioscler Thromb Vasc Biol. 2016. PMID: 27339458
-
Ginsenoside-Rp3 inhibits platelet activation and thrombus formation by regulating MAPK and cyclic nucleotide signaling.Vascul Pharmacol. 2018 Oct;109:45-55. doi: 10.1016/j.vph.2018.06.002. Epub 2018 Jun 8. Vascul Pharmacol. 2018. PMID: 29890296
-
Small-molecule inhibitors of PDK1.ChemMedChem. 2008 Dec;3(12):1810-38. doi: 10.1002/cmdc.200800195. ChemMedChem. 2008. PMID: 18972468 Review.
-
Acute regulation of PDK1 by a complex interplay of molecular switches.Biochem Soc Trans. 2014 Oct;42(5):1435-40. doi: 10.1042/BST20140222. Biochem Soc Trans. 2014. PMID: 25233428 Review.
Cited by
-
Potential mechanism of SARS-CoV-2-associated central and peripheral nervous system impairment.Acta Neurol Scand. 2022 Sep;146(3):225-236. doi: 10.1111/ane.13657. Epub 2022 Jun 14. Acta Neurol Scand. 2022. PMID: 35699161 Free PMC article. Review.
-
Crosstalk between Platelets and SARS-CoV-2: Implications in Thrombo-Inflammatory Complications in COVID-19.Int J Mol Sci. 2023 Sep 15;24(18):14133. doi: 10.3390/ijms241814133. Int J Mol Sci. 2023. PMID: 37762435 Free PMC article. Review.
-
Brevianamide F Exerts Antithrombotic Effects by Modulating the MAPK Signaling Pathway and Coagulation Cascade.Mar Drugs. 2024 Sep 26;22(10):439. doi: 10.3390/md22100439. Mar Drugs. 2024. PMID: 39452847 Free PMC article.
-
MAPK-interacting kinase 1 regulates platelet production, activation, and thrombosis.Blood. 2022 Dec 8;140(23):2477-2489. doi: 10.1182/blood.2022015568. Blood. 2022. PMID: 35930749 Free PMC article.
-
Megakaryocyte-specific knockout of the Mir-99b/let7e/125a cluster lowers platelet count without altering platelet function.Blood Cells Mol Dis. 2021 Dec;92:102624. doi: 10.1016/j.bcmd.2021.102624. Epub 2021 Nov 3. Blood Cells Mol Dis. 2021. PMID: 34775219 Free PMC article.
References
-
- Donati MB. Cancer and thrombosis. Haemostasis. 1994;24:128–31. - PubMed
-
- Mannaioni PF, Di Bello MG, Masini E. Platelets and inflammation: role of platelet-derived growth factor, adhesion molecules and histamine. Inflamm Res. 1997;46:4–18. - PubMed
-
- Goldschmidt-Clermont PJ, Kandzari DE, Sketch MH, Jr, Phillips HR. Inflammation, platelets, and glycoprotein IIb/IIIa inhibitors. J Invasive Cardiol. 2002;14(Suppl E):18E–25E. quiz 6E. - PubMed
-
- Weyrich AS, Lindemann S, Zimmerman GA. The evolving role of platelets in inflammation. J Thromb Haemost. 2003;1:1897–905. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

