Hypoexcitability precedes denervation in the large fast-contracting motor units in two unrelated mouse models of ALS
- PMID: 29580378
- PMCID: PMC5922970
- DOI: 10.7554/eLife.30955
Hypoexcitability precedes denervation in the large fast-contracting motor units in two unrelated mouse models of ALS
Abstract
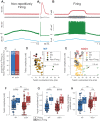
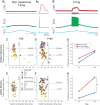
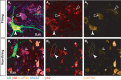
Hyperexcitability has been suggested to contribute to motoneuron degeneration in amyotrophic lateral sclerosis (ALS). If this is so, and given that the physiological type of a motor unit determines the relative susceptibility of its motoneuron in ALS, then one would expect the most vulnerable motoneurons to display the strongest hyperexcitability prior to their degeneration, whereas the less vulnerable should display a moderate hyperexcitability, if any. We tested this hypothesis in vivo in two unrelated ALS mouse models by correlating the electrical properties of motoneurons with their physiological types, identified based on their motor unit contractile properties. We found that, far from being hyperexcitable, the most vulnerable motoneurons become unable to fire repetitively despite the fact that their neuromuscular junctions were still functional. Disease markers confirm that this loss of function is an early sign of degeneration. Our results indicate that intrinsic hyperexcitability is unlikely to be the cause of motoneuron degeneration.
Keywords: electrophysiology; firing properties; in vivo intracellular recordings; motoneuron; motor neuron; mouse; neuroscience.
© 2018, Martínez-Silva et al.
Conflict of interest statement
MM, RI, AS, CH, NS, FR, DZ, MM No competing interests declared
Figures






Comment in
-
Marking the differences in motoneurons.Elife. 2018 Apr 27;7:e36832. doi: 10.7554/eLife.36832. Elife. 2018. PMID: 29701592 Free PMC article.
References
-
- Bernard-Marissal N, Moumen A, Sunyach C, Pellegrino C, Dudley K, Henderson CE, Raoul C, Pettmann B. Reduced calreticulin levels link endoplasmic reticulum stress and Fas-triggered cell death in motoneurons vulnerable to ALS. Journal of Neuroscience. 2012;32:4901–4912. doi: 10.1523/JNEUROSCI.5431-11.2012. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

