The Sealing Zone in Osteoclasts: A Self-Organized Structure on the Bone
- PMID: 29587415
- PMCID: PMC5979552
- DOI: 10.3390/ijms19040984
The Sealing Zone in Osteoclasts: A Self-Organized Structure on the Bone
Abstract
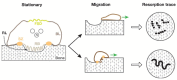
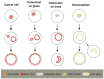
Osteoclasts form a specialized cell-matrix adhesion structure, known as the "sealing zone", during bone resorption. The sealing zone is a dynamic actin-rich structure that defines the resorption area of the bone. The detailed dynamics and fine structure of the sealing zone have been elusive. Osteoclasts plated on glass do not form a sealing zone, but generate a separate supra-molecular structure called the "podosome belt". Podosomes are integrin-based adhesion complexes involved in matrix adhesion, cell migration, matrix degradation, and mechanosensing. Invadopodia, podosome-like protrusions in cancer cells, are involved in cell invasion into other tissues by promoting matrix degradation. Both podosomes and invadopodia exhibit actin pattern transitions during maturation. We previously found that Arp2/3-dependent actin flow occurs in all observed assembly patterns of podosomes in osteoclasts on glass. It is known that the actin wave in Dictyostelium cells exhibits a similar pattern transition in its evolution. Because of significant advances in our understanding regarding the mechanism of podosomes/invadopodia formation over the last decade, we revisited the structure and function of the sealing zone in this review, highlighting the possible involvement of self-organized actin waves in the organogenesis of the sealing zone.
Keywords: Arp2/3; actin polymerization; actin wave; integrin; invadopodia; osteoclasts; plasma membrane; podosome; sealing zone.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

