The Typhoid Toxin Produced by the Nontyphoidal Salmonella enterica Serotype Javiana Is Required for Induction of a DNA Damage Response In Vitro and Systemic Spread In Vivo
- PMID: 29588404
- PMCID: PMC5874915
- DOI: 10.1128/mBio.00467-18
The Typhoid Toxin Produced by the Nontyphoidal Salmonella enterica Serotype Javiana Is Required for Induction of a DNA Damage Response In Vitro and Systemic Spread In Vivo
Abstract
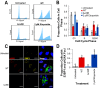
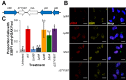
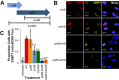
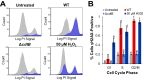
The Salmonella cytolethal distending toxin (S-CDT), first described as the "typhoid toxin" in Salmonella enterica subsp. enterica serotype Typhi, induces DNA damage in eukaryotic cells. Recent studies have shown that more than 40 nontyphoidal Salmonella (NTS) serotypes carry genes that encode S-CDT, yet very little is known about the activity, function, and role of S-CDT in NTS. Here we show that deletion of genes encoding the binding subunit (pltB) and a bacteriophage muramidase predicted to play a role in toxin export (ttsA) does not abolish toxin activity in the S-CDT-positive NTS Salmonella enterica subsp. enterica serotype Javiana. However, S. Javiana strains harboring deletions of both pltB and its homolog artB, had a complete loss of S-CDT activity, suggesting that S. Javiana carries genes encoding two variants of the binding subunit. S-CDT-mediated DNA damage, as determined by phosphorylation of histone 2AX (H2AX), producing phosphorylated H2AX (γH2AX), was restricted to epithelial cells in S and G2/M phases of the cell cycle and did not result in apoptosis or cell death. Compared to mice infected with a ΔcdtB strain, mice infected with wild-type S. Javiana had significantly higher levels of S. Javiana in the liver, but not in the spleen, ileum, or cecum. Overall, we show that production of active S-CDT by NTS serotype S. Javiana requires different genes (cdtB, pltA, and either pltB or artB) for expression of biologically active toxin than those reported for S-CDT production by S. Typhi (cdtB, pltA, pltB, and ttsA). However, as in S. Typhi, NTS S-CDT influences the outcome of infection both in vitro and in vivoIMPORTANCE Nontyphoidal Salmonella (NTS) are a major cause of bacterial food-borne illness worldwide; however, our understanding of virulence mechanisms that determine the outcome and severity of nontyphoidal salmonellosis is incompletely understood. Here we show that S-CDT produced by NTS plays a significant role in the outcome of infection both in vitro and in vivo, highlighting S-CDT as an important virulence factor for nontyphoidal Salmonella serotypes. Our data also contribute novel information about the function of S-CDT, as S-CDT-mediated DNA damage occurs only during certain phases of the cell cycle, and the resulting damage does not induce cell death as assessed using a propidium iodide exclusion assay. Importantly, our data support that, despite having genetically similar S-CDT operons, NTS serotype S. Javiana has different genetic requirements than S. Typhi, for the production and export of active S-CDT.
Keywords: DNA damage; Salmonella; nontyphoidal; typhoid toxin.
Copyright © 2018 Miller et al.
Figures


Similar articles
-
The Majority of Typhoid Toxin-Positive Salmonella Serovars Encode ArtB, an Alternate Binding Subunit.mSphere. 2021 Jan 6;6(1):e01255-20. doi: 10.1128/mSphere.01255-20. mSphere. 2021. PMID: 33408236 Free PMC article.
-
The Cytolethal Distending Toxin Produced by Nontyphoidal Salmonella Serotypes Javiana, Montevideo, Oranienburg, and Mississippi Induces DNA Damage in a Manner Similar to That of Serotype Typhi.mBio. 2016 Dec 20;7(6):e02109-16. doi: 10.1128/mBio.02109-16. mBio. 2016. PMID: 27999166 Free PMC article.
-
Detection and functionality of the CdtB, PltA, and PltB from Salmonella enterica serovar Javiana.Pathog Dis. 2014 Nov;72(2):95-103. doi: 10.1111/2049-632X.12191. Epub 2014 Jul 16. Pathog Dis. 2014. PMID: 24891290
-
Dynamic Duo-The Salmonella Cytolethal Distending Toxin Combines ADP-Ribosyltransferase and Nuclease Activities in a Novel Form of the Cytolethal Distending Toxin.Toxins (Basel). 2016 Apr 25;8(5):121. doi: 10.3390/toxins8050121. Toxins (Basel). 2016. PMID: 27120620 Free PMC article. Review.
-
The contribution of cytolethal distending toxin to bacterial pathogenesis.Crit Rev Microbiol. 2006 Oct-Dec;32(4):227-48. doi: 10.1080/10408410601023557. Crit Rev Microbiol. 2006. PMID: 17123907 Review.
Cited by
-
Recent Advances in the Detection of Antibiotic and Multi-Drug Resistant Salmonella: An Update.Int J Mol Sci. 2021 Mar 28;22(7):3499. doi: 10.3390/ijms22073499. Int J Mol Sci. 2021. PMID: 33800682 Free PMC article. Review.
-
P. aeruginosa interactions with other microbes in biofilms during co-infection.AIMS Microbiol. 2023 Aug 10;9(4):612-646. doi: 10.3934/microbiol.2023032. eCollection 2023. AIMS Microbiol. 2023. PMID: 38173971 Free PMC article. Review.
-
Co- and polymicrobial infections in the gut mucosa: The host-microbiota-pathogen perspective.Cell Microbiol. 2021 Feb;23(2):e13279. doi: 10.1111/cmi.13279. Epub 2020 Oct 22. Cell Microbiol. 2021. PMID: 33040471 Free PMC article. Review.
-
The Majority of Typhoid Toxin-Positive Salmonella Serovars Encode ArtB, an Alternate Binding Subunit.mSphere. 2021 Jan 6;6(1):e01255-20. doi: 10.1128/mSphere.01255-20. mSphere. 2021. PMID: 33408236 Free PMC article.
-
The diverse landscape of AB5-type toxins.Eng Microbiol. 2023 Jun 25;3(4):100104. doi: 10.1016/j.engmic.2023.100104. eCollection 2023 Dec. Eng Microbiol. 2023. PMID: 39628907 Free PMC article. Review.
References
-
- Majowicz SE, Musto J, Scallan E, Angulo FJ, Kirk M, O’Brien SJ, Jones TF, Fazil A, Hoekstra RM, International Collaboration on Enteric Disease “Burden of Illness” Studies . 2010. The global burden of nontyphoidal Salmonella gastroenteritis. Clin Infect Dis 50:882–889. doi: 10.1086/650733. - DOI - PubMed
-
- Kirk MD, Pires SM, Black RE, Caipo M, Crump JA, Devleesschauwer B, Döpfer D, Fazil A, Fischer-Walker CL, Hald T, Hall AJ, Keddy KH, Lake RJ, Lanata CF, Torgerson PR, Havelaar AH, Angulo FJ. 2015. World Health Organization estimates of the global and regional disease burden of 22 foodborne bacterial, protozoal, and viral diseases, 2010: a data synthesis. PLoS Med 12:e1001921. doi: 10.1371/journal.pmed.1001921. - DOI - PMC - PubMed
-
- den Bakker HC, Moreno Switt AI, Govoni G, Cummings CA, Ranieri ML, Degoricija L, Hoelzer K, Rodriguez-Rivera LD, Brown S, Bolchacova E, Furtado MR, Wiedmann M. 2011. Genome sequencing reveals diversification of virulence factor content and possible host adaptation in distinct subpopulations of Salmonella enterica. BMC Genomics 12:425. doi: 10.1186/1471-2164-12-425. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

