Optimization of Cholinesterase-Based Catalytic Bioscavengers Against Organophosphorus Agents
- PMID: 29593539
- PMCID: PMC5859046
- DOI: 10.3389/fphar.2018.00211
Optimization of Cholinesterase-Based Catalytic Bioscavengers Against Organophosphorus Agents
Abstract
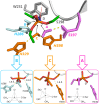
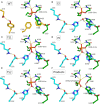
Organophosphorus agents (OPs) are irreversible inhibitors of acetylcholinesterase (AChE). OP poisoning causes major cholinergic syndrome. Current medical counter-measures mitigate the acute effects but have limited action against OP-induced brain damage. Bioscavengers are appealing alternative therapeutic approach because they neutralize OPs in bloodstream before they reach physiological targets. First generation bioscavengers are stoichiometric bioscavengers. However, stoichiometric neutralization requires administration of huge doses of enzyme. Second generation bioscavengers are catalytic bioscavengers capable of detoxifying OPs with a turnover. High bimolecular rate constants (kcat/Km > 106 M-1min-1) are required, so that low enzyme doses can be administered. Cholinesterases (ChE) are attractive candidates because OPs are hemi-substrates. Moderate OP hydrolase (OPase) activity has been observed for certain natural ChEs and for G117H-based human BChE mutants made by site-directed mutagenesis. However, before mutated ChEs can become operational catalytic bioscavengers their dephosphylation rate constant must be increased by several orders of magnitude. New strategies for converting ChEs into fast OPase are based either on combinational approaches or on computer redesign of enzyme. The keystone for rational conversion of ChEs into OPases is to understand the reaction mechanisms with OPs. In the present work we propose that efficient OP hydrolysis can be achieved by re-designing the configuration of enzyme active center residues and by creating specific routes for attack of water molecules and proton transfer. Four directions for nucleophilic attack of water on phosphorus atom were defined. Changes must lead to a novel enzyme, wherein OP hydrolysis wins over competing aging reactions. Kinetic, crystallographic, and computational data have been accumulated that describe mechanisms of reactions involving ChEs. From these studies, it appears that introducing new groups that create a stable H-bonded network susceptible to activate and orient water molecule, stabilize transition states (TS), and intermediates may determine whether dephosphylation is favored over aging. Mutations on key residues (L286, F329, F398) were considered. QM/MM calculations suggest that mutation L286H combined to other mutations favors water attack from apical position. However, the aging reaction is competing. Axial direction of water attack is not favorable to aging. QM/MM calculation shows that F329H+F398H-based multiple mutants display favorable energy barrier for fast reactivation without aging.
Keywords: acetylcholinesterase; bioscavenger; butyrylcholinesterase; computer design; organophosphorus compound; phosphotriesterase.
Figures





Similar articles
-
A collaborative endeavor to design cholinesterase-based catalytic scavengers against toxic organophosphorus esters.Chem Biol Interact. 2008 Sep 25;175(1-3):273-80. doi: 10.1016/j.cbi.2008.04.005. Epub 2008 Apr 16. Chem Biol Interact. 2008. PMID: 18508040
-
Bioscavengers for the protection of humans against organophosphate toxicity.Chem Biol Interact. 2005 Dec 15;157-158:167-71. doi: 10.1016/j.cbi.2005.10.024. Epub 2005 Nov 15. Chem Biol Interact. 2005. PMID: 16293236
-
Catalytic bioscavengers against organophosphorus agents: mechanistic issues of self-reactivating cholinesterases.Toxicology. 2018 Nov 1;409:91-102. doi: 10.1016/j.tox.2018.07.020. Epub 2018 Jul 26. Toxicology. 2018. PMID: 30056174
-
Emergence of catalytic bioscavengers against organophosphorus agents.Chem Biol Interact. 2016 Nov 25;259(Pt B):319-326. doi: 10.1016/j.cbi.2016.02.010. Epub 2016 Feb 17. Chem Biol Interact. 2016. PMID: 26899146 Review.
-
Progress in the development of enzyme-based nerve agent bioscavengers.Chem Biol Interact. 2013 Dec 5;206(3):536-44. doi: 10.1016/j.cbi.2013.06.012. Epub 2013 Jun 26. Chem Biol Interact. 2013. PMID: 23811386 Review.
Cited by
-
QM/MM Description of Newly Selected Catalytic Bioscavengers Against Organophosphorus Compounds Revealed Reactivation Stimulus Mediated by Histidine Residue in the Acyl-Binding Loop.Front Pharmacol. 2018 Aug 3;9:834. doi: 10.3389/fphar.2018.00834. eCollection 2018. Front Pharmacol. 2018. PMID: 30123127 Free PMC article.
-
Organophosphorus Nerve Agents: Types, Toxicity, and Treatments.J Toxicol. 2020 Sep 22;2020:3007984. doi: 10.1155/2020/3007984. eCollection 2020. J Toxicol. 2020. PMID: 33029136 Free PMC article. Review.
-
Acetylcholinesterase: The "Hub" for Neurodegenerative Diseases and Chemical Weapons Convention.Biomolecules. 2020 Mar 7;10(3):414. doi: 10.3390/biom10030414. Biomolecules. 2020. PMID: 32155996 Free PMC article. Review.
-
Positron emission tomography evaluation of oxime countermeasures in live rats using the tracer O-(2-[18 F]fluoroethyl)-O-(p-nitrophenyl)methylphosphonate [18 F]-VXS.Ann N Y Acad Sci. 2020 Nov;1479(1):180-195. doi: 10.1111/nyas.14363. Epub 2020 May 20. Ann N Y Acad Sci. 2020. PMID: 32436233 Free PMC article.
-
Interaction of Cucurbit[7]uril with Oxime K027, Atropine, and Paraoxon: Risky or Advantageous Delivery System?Int J Mol Sci. 2020 Oct 23;21(21):7883. doi: 10.3390/ijms21217883. Int J Mol Sci. 2020. PMID: 33114215 Free PMC article.
References
-
- Albaret C., Masson P., Broomfield C. A., El Kaim L., Fortier P. L. (1998). Mechanical aspects of the phosphotriesterase activity of human butyrylcholinesterase G117H mutant, in Structure and Function of Cholinesterases and Related Proteins, 1st Edn., eds Doctor B. P., Quinn D. M., Taylot P., Rotundo R. L. (New York, NY: Plenum Press; ), 399–405.
-
- Ashani Y., Leader H., Aggarwal N., Silman I., Worek F., Sussman J. L., et al. . (2016). In vitro evaluation of the catalytic activity of paraoxonases and phosphotriesterases predicts the enzyme circulatory levels required for in vivo protection against organophosphate intoxications. Chem. Biol. Interact. 259(Pt B), 252–256. 10.1016/j.cbi.2016.04.039 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

