Treatment of Arsenite Intoxication-Induced Peripheral Vasculopathy with Mesenchymal Stem Cells
- PMID: 29596344
- PMCID: PMC5979449
- DOI: 10.3390/ijms19041026
Treatment of Arsenite Intoxication-Induced Peripheral Vasculopathy with Mesenchymal Stem Cells
Abstract
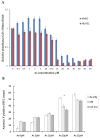
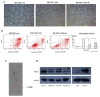
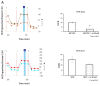
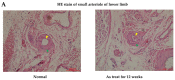
Arsenite (As), a notorious toxic metal, is ubiquitously distributed in the earth and poses a serious threat to human health. Histopathological lesions of As intoxication are known as thromboangiitis obliterans, which are resistant to current treatment and often lead to lower limb amputation. In this study, we attempt to find that treatment with mesenchymal stem cells (MSCs) may be effective for As-induced vasculopathy. We first conducted an in vitro study with a co-culture system containing human MSCs and human umbilical vein endothelial cells (HUVECs) and treated individual and co-cultured cells with various concentrations of arsenite. We also designed an in vivo study in which Sprague Dawley (SD) rats received periodic intraperitoneal (IP) injections of 16 ppm arsenite for 12 weeks. MSCs were harvested from BALB/c mice that were transplanted via tail vein injection. We found that there was significantly higher cellular viability in human mesenchymal stem cells (hMSCs) than in HUVECs under concentrations of arsenite between 15 and 25 μM. The Annexin V apoptosis assay further confirmed this finding. Cytokine array assay for As-conditioned media revealed an elevated vascular endothelial growth factor (VEGF) level secreted by MSCs, which is crucial for HUVEC survival and was evaluated by an siRNA VEGF knockdown test. In the in vivo study, we demonstrated early apoptotic changes in the anterior tibial vessels of As-injected SD rats with a Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay, but these apoptotic changes were less frequently observed upon MSCs transplantation, indicating that the cytoprotective effect of MSCs successfully protected against As-induced peripheral vasculopathy. The feasibility of MSCs to treat and /or prevent the progression of As-induced vasculopathy is justified. Further clinical studies are required to demonstrate the therapeutic efficacy of MSCs in patients suffering from As intoxication with vasculopathy.
Keywords: arsenite; mesenchymal stem cells; peripheral vascular disease.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Wu M.M., Chiou H.Y., Wang T.W., Hsueh Y.M., Wang I.H., Chen C.J., Lee T.C. Association of blood arsenic levels with increased reactive oxidants and decreased antioxidant capacity in a human population of northeastern Taiwan. Environ. Health Perspect. 2001;109:1011–1017. doi: 10.1289/ehp.011091011. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

