Akkermansia muciniphila is permissive to arthritis in the K/BxN mouse model of arthritis
- PMID: 29599513
- PMCID: PMC6153082
- DOI: 10.1038/s41435-018-0024-1
Akkermansia muciniphila is permissive to arthritis in the K/BxN mouse model of arthritis
Abstract
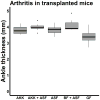
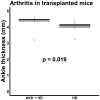
Studies have identified abnormalities in the microbiota of patients with arthritis. To evaluate the pathogenicity of human microbiota, we performed fecal microbial transplantation from children with spondyloarthritis and controls to germ-free KRN/B6xNOD mice. Ankle swelling was equivalent in those that received patient vs. control microbiota. Principal coordinates analysis revealed incomplete uptake of the human microbiota with over-representation of two genera (Bacteroides and Akkermansia) among the transplanted mice. The microbiota predicted the extent of ankle swelling (R2 = 0.185, p = 0.018). The abundances of Bacteroides (r = -0.510, p = 0.010) inversely and Akkermansia (r = 0.367, p = 0.078) directly correlated with ankle swelling. Addition of Akkermansia muciniphila to Altered Schaedler's Flora (ASF) resulted in small but statistically significant increased ankle swelling as compared to mice that received ASF alone (4.0 mm, 3.9-4.1 vs. 3.9 mm, IQR 3.6-4.0, p = 0.041), as did addition of A. muciniphila cultures to transplanted human microbiota as compared to mice that received transplanted human microbiota alone (4.5 mm, IQR 4.3-5.5 vs. 4.1 mm, IQR 3.9-4.3, p = 0.019). This study supports previous findings of an association between A. muciniphila and arthritis.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures


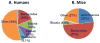


Similar articles
-
Akkermansia muciniphila induces gut microbiota remodelling and controls islet autoimmunity in NOD mice.Gut. 2018 Aug;67(8):1445-1453. doi: 10.1136/gutjnl-2017-314508. Epub 2017 Dec 21. Gut. 2018. PMID: 29269438
-
Effect of inulin, galacto-oligosaccharides, and polyphenols on the gut microbiota, with a focus on Akkermansia muciniphila.Food Funct. 2024 May 7;15(9):4763-4772. doi: 10.1039/d4fo00428k. Food Funct. 2024. PMID: 38590256
-
An Open-Labeled Study on Fecal Microbiota Transfer in Irritable Bowel Syndrome Patients Reveals Improvement in Abdominal Pain Associated with the Relative Abundance of Akkermansia Muciniphila.Digestion. 2019;100(2):127-138. doi: 10.1159/000494252. Epub 2018 Nov 13. Digestion. 2019. PMID: 30423561
-
Action and function of Akkermansia muciniphila in microbiome ecology, health and disease.Best Pract Res Clin Gastroenterol. 2017 Dec;31(6):637-642. doi: 10.1016/j.bpg.2017.10.001. Epub 2017 Oct 13. Best Pract Res Clin Gastroenterol. 2017. PMID: 29566906 Review.
-
The metabolic, protective, and immune functions of Akkermansia muciniphila.Microbiol Res. 2023 Jan;266:127245. doi: 10.1016/j.micres.2022.127245. Epub 2022 Oct 28. Microbiol Res. 2023. PMID: 36347103 Review.
Cited by
-
Putative Pathobionts in HLA-B27-Associated Spondyloarthropathy.Front Immunol. 2021 Jan 18;11:586494. doi: 10.3389/fimmu.2020.586494. eCollection 2020. Front Immunol. 2021. PMID: 33537028 Free PMC article. Review.
-
Consistent Alterations of Human Fecal Microbes After Transplantation into Germ-free Mice.Genomics Proteomics Bioinformatics. 2022 Apr;20(2):382-393. doi: 10.1016/j.gpb.2020.06.024. Epub 2021 Jun 9. Genomics Proteomics Bioinformatics. 2022. PMID: 34118462 Free PMC article.
-
An Association of Gut Microbiota with Different Phenotypes in Chinese Patients with Rheumatoid Arthritis.J Clin Med. 2019 Oct 24;8(11):1770. doi: 10.3390/jcm8111770. J Clin Med. 2019. PMID: 31652955 Free PMC article.
-
Akkermansia muciniphila as a Next-Generation Probiotic in Modulating Human Metabolic Homeostasis and Disease Progression: A Role Mediated by Gut-Liver-Brain Axes?Int J Mol Sci. 2023 Feb 15;24(4):3900. doi: 10.3390/ijms24043900. Int J Mol Sci. 2023. PMID: 36835309 Free PMC article. Review.
-
Safety of pasteurised Akkermansia muciniphila as a novel food pursuant to Regulation (EU) 2015/2283.EFSA J. 2021 Sep 1;19(9):e06780. doi: 10.2903/j.efsa.2021.6780. eCollection 2021 Sep. EFSA J. 2021. PMID: 34484452 Free PMC article.
References
-
- Tejesvi MV, Arvonen M, Kangas SM, Keskitalo PL, Pirttila AM, Karttunen TJ, et al. Faecal microbiome in new-onset juvenile idiopathic arthritis. Eur J Clin Microbiol Infect Dis. 2016;35(3):363–70. - PubMed
-
- Maeda Y, Kurakawa T, Umemoto E, Motooka D, Ito Y, Gotoh K, et al. Dysbiosis contributes to arthritis development via activation of autoreactive T cells in the intestine. Arthritis Rheumatol. 2016 - PubMed
-
- Derrien M, Vaughan EE, Plugge CM, de Vos WM. Akkermansia muciniphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int J Syst Evol Microbiol. 2004;54(Pt 5):1469–76. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

