Expression of novel "LOCGEF" isoforms of ARHGEF18 in eosinophils
- PMID: 29601110
- PMCID: PMC6013370
- DOI: 10.1002/JLB.2MA1017-418RR
Expression of novel "LOCGEF" isoforms of ARHGEF18 in eosinophils
Abstract
Genomic, transcriptomic and proteomic databases indicate that the N-terminal 322 residues encoded by the presumptive LOC100996504 gene, which is adjacent to the ARHGEF18 guanine nucleotide exchange factor gene on chromosome 19, constitute the N-terminal portion of a 1361-residue isoform of ARHGEF18, dubbed LOCGEF-X3. LOCGEF-X3 arises from the use of a leukocyte-specific alternative transcriptional start site and splicing that bypasses the initial noncoding exon of the canonical 1015-residue ARHGEF18 isoform, p114. Eosinophil LOCGEF-X3 was amplified and cloned, recombinant LOCGEF-X3 was expressed, and anti-ARHGEF18 antibody was found to recognize a band in immunoblots of eosinophil lysates that co-migrates with recombinant LOCGEF-X3. PCR of eosinophils revealed minor amounts of transcripts for X4 and X5 isoforms of LOCGEF that arise from differential splicing and differ from the X3 isoform at their extreme N-termini. No p114 transcript or protein band was detected in eosinophils. Immunostaining with anti-ARHGEF18 antibody revealed relocalization of LOCGEF and RHOA from the periphery of round unstimulated eosinophils to the 2 poles of eosinophils polarized by treatment with IL5, CCL11, or IL33 in suspension. Canonical p114 ARHGEF18 has been implicated in maintenance of epithelial cell polarity. We suggest that the "LOC" portion of LOCGEF, which is unlike any other protein domain, has unique functions in control of polarity in activated eosinophils and other leukocytes.
Keywords: ARHGEF18; LOC100996504; alternative transcriptional start; granulocyte; guanine nucleotide exchange factor; p114-GEF.
©2018 Society for Leukocyte Biology.
Conflict of interest statement
Conflicts of interest disclosure
The authors declare no conflict of interest.
Figures

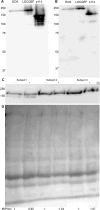

Similar articles
-
ArhGEF18 regulates RhoA-Rock2 signaling to maintain neuro-epithelial apico-basal polarity and proliferation.Development. 2013 Jul;140(13):2787-97. doi: 10.1242/dev.096487. Epub 2013 May 22. Development. 2013. PMID: 23698346
-
ARHGEF18 can promote BVDV NS5B activation of the host NF-κB signaling pathway by combining with the NS5B-palm domain.Vet Microbiol. 2024 Apr;291:109911. doi: 10.1016/j.vetmic.2023.109911. Epub 2023 Nov 7. Vet Microbiol. 2024. PMID: 38367539
-
Establishment and functional studies of a model of cardiomyopathy with cardiomyocyte-specific conditional knockout of Arhgef18.Dis Model Mech. 2025 Mar 1;18(3):dmm052172. doi: 10.1242/dmm.052172. Epub 2025 Mar 31. Dis Model Mech. 2025. PMID: 40159883 Free PMC article.
-
IL-5 and IL-5 receptor in asthma.Mem Inst Oswaldo Cruz. 1997;92 Suppl 2:75-91. doi: 10.1590/s0074-02761997000800012. Mem Inst Oswaldo Cruz. 1997. PMID: 9698919 Review.
-
The guanine nucleotide exchange factor Tiam1: a Janus-faced molecule in cellular signaling.Cell Signal. 2014 Mar;26(3):483-91. doi: 10.1016/j.cellsig.2013.11.034. Epub 2013 Dec 2. Cell Signal. 2014. PMID: 24308970 Review.
Cited by
-
GPCRs that Rhoar the Guanine nucleotide exchange factors.Small GTPases. 2022 Jan;13(1):84-99. doi: 10.1080/21541248.2021.1896963. Epub 2021 Apr 14. Small GTPases. 2022. PMID: 33849392 Free PMC article. Review.
-
Revealing potential diagnostic gene biomarkers of septic shock based on machine learning analysis.BMC Infect Dis. 2022 Jan 19;22(1):65. doi: 10.1186/s12879-022-07056-4. BMC Infect Dis. 2022. PMID: 35045818 Free PMC article.
References
-
- Niu J, Profirovic J, Pan H, Vaiskunaite R, Voyno-Yasenetskaya T. G Protein betagamma subunits stimulate p114RhoGEF, a guanine nucleotide exchange factor for RhoA and Rac1: regulation of cell shape and reactive oxygen species production. Circ Res. 2003;93:848–56. - PubMed
-
- Herder C, Swiercz JM, Muller C, Peravali R, Quiring R, Offermanns S, Wittbrodt J, Loosli F. ArhGEF18 regulates RhoA-Rock2 signaling to maintain neuro-epithelial apico-basal polarity and proliferation. Development. 2013;140:2787–97. - PubMed
-
- Nagata K, Inagaki M. Cytoskeletal modification of Rho guanine nucleotide exchange factor activity: identification of a Rho guanine nucleotide exchange factor as a binding partner for Sept9b, a mammalian septin. Oncogene. 2005;24:65–76. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

