Oversized galactosides as a probe for conformational dynamics in LacY
- PMID: 29602806
- PMCID: PMC5910868
- DOI: 10.1073/pnas.1800706115
Oversized galactosides as a probe for conformational dynamics in LacY
Abstract
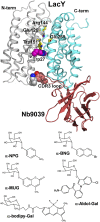
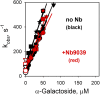
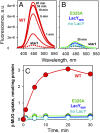
Binding kinetics of α-galactopyranoside homologs with fluorescent aglycones of different sizes and shapes were determined with the lactose permease (LacY) of Escherichia coli by FRET from Trp151 in the binding site of LacY to the fluorophores. Fast binding was observed with LacY stabilized in an outward-open conformation (kon = 4-20 μM-1·s-1), indicating unobstructed access to the binding site even for ligands that are much larger than lactose. Dissociation rate constants (koff) increase with the size of the aglycone so that Kd values also increase but remain in the micromolar range for each homolog. Phe27 (helix I) forms an apparent constriction in the pathway for sugar by protruding into the periplasmic cavity. However, replacement of Phe27 with a bulkier Trp does not create an obstacle in the pathway even for large ligands, since binding kinetics remain unchanged. High accessibility of the binding site is also observed in a LacY/nanobody complex with partially blocked periplasmic opening. Remarkably, E. coli expressing WT LacY catalyzes transport of α- or β-galactopyranosides with oversized aglycones such as bodipy or Aldol518, which may require an extra space within the occluded intermediate. The results confirm that LacY specificity is strictly directed toward the galactopyranoside ring and also clearly indicate that the opening on the periplasmic side is sufficiently wide to accommodate the large galactoside derivatives tested here. We conclude that the actual pathway for the substrate entering from the periplasmic side is wider than the pore diameter calculated in the periplasmic-open X-ray structures.
Keywords: fluorescence; lactose permease; membrane transport proteins; nanobodies; stopped-flow.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Engineered occluded apo-intermediate of LacY.Proc Natl Acad Sci U S A. 2018 Dec 11;115(50):12716-12721. doi: 10.1073/pnas.1816267115. Epub 2018 Nov 26. Proc Natl Acad Sci U S A. 2018. PMID: 30478058 Free PMC article.
-
Outward-facing conformers of LacY stabilized by nanobodies.Proc Natl Acad Sci U S A. 2014 Dec 30;111(52):18548-53. doi: 10.1073/pnas.1422265112. Epub 2014 Dec 15. Proc Natl Acad Sci U S A. 2014. PMID: 25512549 Free PMC article.
-
Crystal structure of a LacY-nanobody complex in a periplasmic-open conformation.Proc Natl Acad Sci U S A. 2016 Nov 1;113(44):12420-12425. doi: 10.1073/pnas.1615414113. Epub 2016 Oct 19. Proc Natl Acad Sci U S A. 2016. PMID: 27791182 Free PMC article.
-
Lessons from lactose permease.Annu Rev Biophys Biomol Struct. 2006;35:67-91. doi: 10.1146/annurev.biophys.35.040405.102005. Annu Rev Biophys Biomol Struct. 2006. PMID: 16689628 Free PMC article. Review.
-
The alternating access transport mechanism in LacY.J Membr Biol. 2011 Jan;239(1-2):85-93. doi: 10.1007/s00232-010-9327-5. Epub 2010 Dec 16. J Membr Biol. 2011. PMID: 21161516 Free PMC article. Review.
Cited by
-
Function Trumps Form in Two Sugar Symporters, LacY and vSGLT.Int J Mol Sci. 2021 Mar 30;22(7):3572. doi: 10.3390/ijms22073572. Int J Mol Sci. 2021. PMID: 33808202 Free PMC article. Review.
-
Engineered occluded apo-intermediate of LacY.Proc Natl Acad Sci U S A. 2018 Dec 11;115(50):12716-12721. doi: 10.1073/pnas.1816267115. Epub 2018 Nov 26. Proc Natl Acad Sci U S A. 2018. PMID: 30478058 Free PMC article.
-
It takes two to tango: The dance of the permease.J Gen Physiol. 2019 Jul 1;151(7):878-886. doi: 10.1085/jgp.201912377. Epub 2019 May 30. J Gen Physiol. 2019. PMID: 31147449 Free PMC article. Review.
-
Directed evolution of an α1,3-fucosyltransferase using a single-cell ultrahigh-throughput screening method.Sci Adv. 2019 Oct 9;5(10):eaaw8451. doi: 10.1126/sciadv.aaw8451. eCollection 2019 Oct. Sci Adv. 2019. PMID: 31633018 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

