Cerebrospinal fluid α-synuclein contributes to the differential diagnosis of Alzheimer's disease
- PMID: 29604263
- PMCID: PMC6097943
- DOI: 10.1016/j.jalz.2018.02.015
Cerebrospinal fluid α-synuclein contributes to the differential diagnosis of Alzheimer's disease
Abstract
Introduction: The ability of Alzheimer's disease (AD) cerebrospinal fluid (CSF) biomarkers (amyloid β peptide 1-42, total tau, and phosphorylated tau) to discriminate AD from related disorders is limited. Biomarkers for other concomitant pathologies (e.g., CSF α-synuclein [α-syn] for Lewy body pathology) may be needed to further improve the differential diagnosis.
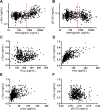
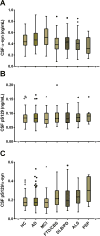
Methods: CSF total α-syn, phosphorylated α-syn at Ser129, and AD CSF biomarkers were evaluated with Luminex immunoassays in 367 participants, followed by validation in 74 different neuropathologically confirmed cases.
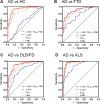
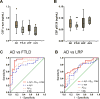
Results: CSF total α-syn, when combined with amyloid β peptide 1-42 and either total tau or phosphorylated tau, improved the differential diagnosis of AD versus frontotemporal dementia, Lewy body disorders, or other neurological disorders. The diagnostic accuracy of the combined models attained clinical relevance (area under curve ∼0.9) and was largely validated in neuropathologically confirmed cases.
Discussion: Combining CSF biomarkers representing AD and Lewy body pathologies may have clinical value in the differential diagnosis of AD.
Keywords: Alzheimer's disease; Biomarkers; Cerebrospinal fluid; Differential diagnosis; α-synuclein.
Copyright © 2018 the Alzheimer's Association. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures
References
-
- McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR, Jr, Kawas CH, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011;7:263–9. - PMC - PubMed
-
- Blennow K, Hampel H, Weiner M, Zetterberg H. Cerebrospinal fluid and plasma biomarkers in Alzheimer disease. Nat Rev Neurol. 2010;6:131–44. - PubMed
-
- Mattsson N, Lonneborg A, Boccardi M, Blennow K, Hansson O. Clinical validity of cerebrospinal fluid Abeta42, tau, and phospho-tau as biomarkers for Alzheimer's disease in the context of a structured 5-phase development framework. Neurobiol Aging. 2017;52:196–213. - PubMed
-
- Ewers M, Mattsson N, Minthon L, Molinuevo JL, Antonell A, Popp J, et al. CSF biomarkers for the differential diagnosis of Alzheimer's disease: A large-scale international multicenter study. Alzheimers Dement. 2015;11:1306–15. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P30 AG010124/AG/NIA NIH HHS/United States
- P01 AG017586/AG/NIA NIH HHS/United States
- R56 AG057417/AG/NIA NIH HHS/United States
- U01 NS091272/NS/NINDS NIH HHS/United States
- P50 NS053488/NS/NINDS NIH HHS/United States
- R21 NS104511/NS/NINDS NIH HHS/United States
- R21 NS085425/NS/NINDS NIH HHS/United States
- U01 NS082137/NS/NINDS NIH HHS/United States
- R21 MH118160/MH/NIMH NIH HHS/United States
- U01 NS097056/NS/NINDS NIH HHS/United States
- R01 AG056711/AG/NIA NIH HHS/United States
- K23 NS088341/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

