Induction of a central memory and stem cell memory phenotype in functionally active CD4+ and CD8+ CAR T cells produced in an automated good manufacturing practice system for the treatment of CD19+ acute lymphoblastic leukemia
- PMID: 29605883
- PMCID: PMC11028239
- DOI: 10.1007/s00262-018-2155-7
Induction of a central memory and stem cell memory phenotype in functionally active CD4+ and CD8+ CAR T cells produced in an automated good manufacturing practice system for the treatment of CD19+ acute lymphoblastic leukemia
Abstract
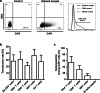
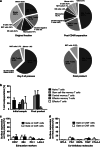
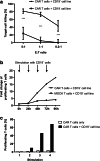
Relapsed/refractory B-precursor acute lymphoblastic leukemia (pre-B ALL) remains a major therapeutic challenge. Chimeric antigen receptor (CAR) T cells are promising treatment options. Central memory T cells (Tcm) and stem cell-like memory T cells (Tscm) are known to promote sustained proliferation and persistence after T-cell therapy, constituting essential preconditions for treatment efficacy. Therefore, we set up a protocol for anti-CD19 CAR T-cell generation aiming at high Tcm/Tscm numbers. 100 ml peripheral blood from pediatric pre-B ALL patients was processed including CD4+/CD8+-separation, T-cell activation with modified anti-CD3/-CD28 reagents and transduction with a 4-1BB-based second generation CAR lentiviral vector. The process was performed on a closed, automated device requiring additional manual/open steps under clean room conditions. The clinical situation of these critically ill and refractory patients with leukemia leads to inconsistent cellular compositions at start of the procedure including high blast counts and low T-cell numbers with exhausted phenotype. Nevertheless, a robust T-cell product was achieved (mean CD4+ = 50%, CD8+ = 39%, transduction = 27%, Tcm = 50%, Tscm = 46%). Strong proliferative potential (up to > 100-fold), specific cytotoxicity and low expression of co-inhibitory molecules were documented. CAR T cells significantly released TH1 cytokines IFN-γ, TNF-α and IL-2 upon target-recognition. In conclusion, partly automated GMP-generation of CAR T cells from critically small blood samples was feasible with a new stimulation protocol that leads to high functionality and expansion potential, balanced CD4/CD8 ratios and a conversion to a Tcm/Tscm phenotype.
Keywords: CAR T cells; GMP production; Pediatric ALL; Tscm/cm.
Conflict of interest statement
Andrew Didier Kaiser and Mario Assenmacher are employees of Miltenyi Biotec. This work has been performed as a collaboration between Tobias Feuchtinger, Franziska Blaeschke and Miltenyi Biotec. Miltenyi Biotec provided reagent free of charge. All other authors declare that they have no conflict of interest.
Figures

 ) procedures are indicated in the lower part of the figure. Cells are cultured in safety level S2. Due to multiple washing and dilution steps, the final product is downscaled to safety level S1. PBMC peripheral blood mononuclear cells, PB peripheral blood, CAR chimeric antigen receptor, GMP good manufacturing practice
) procedures are indicated in the lower part of the figure. Cells are cultured in safety level S2. Due to multiple washing and dilution steps, the final product is downscaled to safety level S1. PBMC peripheral blood mononuclear cells, PB peripheral blood, CAR chimeric antigen receptor, GMP good manufacturing practice

Similar articles
-
Phenotypic and functional attributes of lentivirus-modified CD19-specific human CD8+ central memory T cells manufactured at clinical scale.J Immunother. 2012 Nov-Dec;35(9):689-701. doi: 10.1097/CJI.0b013e318270dec7. J Immunother. 2012. PMID: 23090078 Free PMC article.
-
Generation of clinical-grade CD19-specific CAR-modified CD8+ memory stem cells for the treatment of human B-cell malignancies.Blood. 2016 Jul 28;128(4):519-28. doi: 10.1182/blood-2015-11-683847. Epub 2016 May 25. Blood. 2016. PMID: 27226436 Free PMC article.
-
Closed-system manufacturing of CD19 and dual-targeted CD20/19 chimeric antigen receptor T cells using the CliniMACS Prodigy device at an academic medical center.Cytotherapy. 2018 Mar;20(3):394-406. doi: 10.1016/j.jcyt.2017.09.005. Epub 2017 Dec 26. Cytotherapy. 2018. PMID: 29287970
-
Systematic Review and Meta-analysis of CD19-Specific CAR-T Cell Therapy in Relapsed/Refractory Acute Lymphoblastic Leukemia in the Pediatric and Young Adult Population: Safety and Efficacy Outcomes.Clin Lymphoma Myeloma Leuk. 2021 Apr;21(4):e334-e347. doi: 10.1016/j.clml.2020.12.010. Epub 2020 Dec 17. Clin Lymphoma Myeloma Leuk. 2021. PMID: 33573914
-
Chimeric Antigen Receptor T Cell Therapy for Pediatric B-ALL: Narrowing the Gap Between Early and Long-Term Outcomes.Front Immunol. 2020 Aug 11;11:1985. doi: 10.3389/fimmu.2020.01985. eCollection 2020. Front Immunol. 2020. PMID: 32849662 Free PMC article. Review.
Cited by
-
Transcriptional states of CAR-T infusion relate to neurotoxicity - lessons from high-resolution single-cell SOM expression portraying.Front Immunol. 2022 Sep 28;13:994885. doi: 10.3389/fimmu.2022.994885. eCollection 2022. Front Immunol. 2022. PMID: 36248848 Free PMC article.
-
Cytotoxic Efficiency of Human CD8+ T Cell Memory Subtypes.Front Immunol. 2022 Apr 13;13:838484. doi: 10.3389/fimmu.2022.838484. eCollection 2022. Front Immunol. 2022. PMID: 35493468 Free PMC article.
-
Functionality and Cell Senescence of CD4/ CD8-Selected CD20 CAR T Cells Manufactured Using the Automated CliniMACS Prodigy® Platform.Transfus Med Hemother. 2019 Feb;46(1):47-54. doi: 10.1159/000495772. Epub 2019 Feb 4. Transfus Med Hemother. 2019. PMID: 31244581 Free PMC article. Review.
-
CAR T Cells for Solid Tumors: New Strategies for Finding, Infiltrating, and Surviving in the Tumor Microenvironment.Front Immunol. 2019 Feb 5;10:128. doi: 10.3389/fimmu.2019.00128. eCollection 2019. Front Immunol. 2019. PMID: 30804938 Free PMC article. Review.
-
Harnessing novel strategies and cell types to overcome immune tolerance during adoptive cell therapy in cancer.J Immunother Cancer. 2023 Apr;11(4):e006434. doi: 10.1136/jitc-2022-006434. J Immunother Cancer. 2023. PMID: 37100458 Free PMC article. Review.
References
-
- Blaeschke F, Kaeuferle T, Feucht J, Weber D, Lotfi R, Kaiser A, Assenmacher M, Doering M, Feuchtinger T (2016) Defined central memory and stem memory T cell phenotype of CD4 and CD8 CAR T cells for the treatment of CD19+ acute lymphoblastic leukemia in an automated closed system. In: 58th ASH annual meeting and exposition. Blood, 128(22) (Abstract 4558)
-
- Stenger D, Blaeschke F, Kaeuferle T, Willier S, Lotfi R, Kaiser A, Assenmacher M, Doering M, Feucht J, Feuchtinger T (2017) Automated generation of central memory and stem cell-like memory CD19-specific CAR T cells in a closed, GMP compatible system. In: Cellular therapy, international symposium Erlangen, Erlangen (Abstract 134)
-
- Blaeschke F, Stenger D, Kaeuferle T, Willier S, Lotfi R, Kaiser A, Assenmacher M, Doering M, Feucht J, Feuchtinger T (2017) CD19-specific CAR T cells with a central memory and stem memory phenotype—automated generation in a closed, GMP-compatible system from peripheral blood of pediatric patients with acute lymphoblastic leukemia. In: 43rd annual meeting of the European Society for Blood and Marrow Transplantation (EBMT), Marseille (Abstract)
-
- Blaeschke F, Stenger D, Kaeuferle T, Willier S, Lotfi R, Kaiser A, Assenmacher M, Doering M, Feucht J, Feuchtinger T (2017) Induction of a central memory and stem cell memory phenotype in functionally active CD4+ and CD8+ CAR T cells produced in an automated GMP system for the treatment of CD19+ acute lymphoblastic leukemia. In: Annual meeting of Paediatrische Arbeitsgemeinschaft fuer Stammzelltransplantation and Zelltherapie (PAS&ZT), Hamburg (Abstract) - PMC - PubMed
-
- Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA, Fry TJ, Orentas R, Sabatino M, Shah NN, Steinberg SM, Stroncek D, Tschernia N, Yuan C, Zhang H, Zhang L, Rosenberg SA, Wayne AS, Mackall CL. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 2015;385(9967):517–528. doi: 10.1016/S0140-6736(14)61403-3. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

