Functional Studies of Missense TREM2 Mutations in Human Stem Cell-Derived Microglia
- PMID: 29606617
- PMCID: PMC5998752
- DOI: 10.1016/j.stemcr.2018.03.003
Functional Studies of Missense TREM2 Mutations in Human Stem Cell-Derived Microglia
Abstract
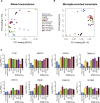
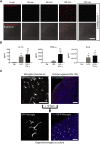
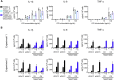
The derivation of microglia from human stem cells provides systems for understanding microglial biology and enables functional studies of disease-causing mutations. We describe a robust method for the derivation of human microglia from stem cells, which are phenotypically and functionally comparable with primary microglia. We used stem cell-derived microglia to study the consequences of missense mutations in the microglial-expressed protein triggering receptor expressed on myeloid cells 2 (TREM2), which are causal for frontotemporal dementia-like syndrome and Nasu-Hakola disease. We find that mutant TREM2 accumulates in its immature form, does not undergo typical proteolysis, and is not trafficked to the plasma membrane. However, in the absence of plasma membrane TREM2, microglia differentiate normally, respond to stimulation with lipopolysaccharide, and are phagocytically competent. These data indicate that dementia-associated TREM2 mutations have subtle effects on microglia biology, consistent with the adult onset of disease in individuals with these mutations.
Keywords: Nasu-Hakola disease; TREM2; dementia; frontotemporal dementia; iPSC-microglia; microglia; neuroinflammation.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

