Site-Directed Spin Labeling EPR for Studying Membrane Proteins
- PMID: 29607317
- PMCID: PMC5828257
- DOI: 10.1155/2018/3248289
Site-Directed Spin Labeling EPR for Studying Membrane Proteins
Abstract
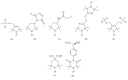
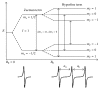
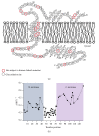
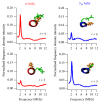
Site-directed spin labeling (SDSL) in combination with electron paramagnetic resonance (EPR) spectroscopy is a rapidly expanding powerful biophysical technique to study the structural and dynamic properties of membrane proteins in a native environment. Membrane proteins are responsible for performing important functions in a wide variety of complicated biological systems that are responsible for the survival of living organisms. In this review, a brief introduction of the most popular SDSL EPR techniques and illustrations of recent applications for studying pertinent structural and dynamic properties on membrane proteins will be discussed.
Figures

Similar articles
-
Electron Paramagnetic Resonance as a Tool for Studying Membrane Proteins.Biomolecules. 2020 May 13;10(5):763. doi: 10.3390/biom10050763. Biomolecules. 2020. PMID: 32414134 Free PMC article. Review.
-
Characterization of the Human KCNQ1 Voltage Sensing Domain (VSD) in Lipodisq Nanoparticles for Electron Paramagnetic Resonance (EPR) Spectroscopic Studies of Membrane Proteins.J Phys Chem B. 2020 Mar 26;124(12):2331-2342. doi: 10.1021/acs.jpcb.9b11506. Epub 2020 Mar 16. J Phys Chem B. 2020. PMID: 32130007 Free PMC article.
-
Site-directed spin labeling EPR spectroscopy in protein research.Biol Chem. 2013 Oct;394(10):1281-300. doi: 10.1515/hsz-2013-0155. Biol Chem. 2013. PMID: 23912220 Review.
-
Elucidating the design principles of photosynthetic electron-transfer proteins by site-directed spin labeling EPR spectroscopy.Biochim Biophys Acta. 2016 May;1857(5):548-556. doi: 10.1016/j.bbabio.2015.08.009. Epub 2015 Sep 1. Biochim Biophys Acta. 2016. PMID: 26334844 Review.
-
EPR Techniques to Probe Insertion and Conformation of Spin-Labeled Proteins in Lipid Bilayers.Methods Mol Biol. 2019;2003:493-528. doi: 10.1007/978-1-4939-9512-7_21. Methods Mol Biol. 2019. PMID: 31218631
Cited by
-
Experimental Validation of the ALLNOX Program for Studying Protein-Nucleic Acid Complexes.J Phys Chem A. 2019 Apr 25;123(16):3592-3598. doi: 10.1021/acs.jpca.9b01027. Epub 2019 Apr 12. J Phys Chem A. 2019. PMID: 30978022 Free PMC article.
-
Lipid Membrane Mimetics in Functional and Structural Studies of Integral Membrane Proteins.Membranes (Basel). 2021 Sep 3;11(9):685. doi: 10.3390/membranes11090685. Membranes (Basel). 2021. PMID: 34564502 Free PMC article. Review.
-
A 13 C-Labeled Triarylmethyl Radical as an EPR Spin Probe Highly Sensitive to Molecular Tumbling.Angew Chem Int Ed Engl. 2020 Sep 14;59(38):16451-16454. doi: 10.1002/anie.202006591. Epub 2020 Jul 21. Angew Chem Int Ed Engl. 2020. PMID: 32542924 Free PMC article.
-
Conformational Differences Are Observed for the Active and Inactive Forms of Pinholin S21 Using DEER Spectroscopy.J Phys Chem B. 2020 Dec 17;124(50):11396-11405. doi: 10.1021/acs.jpcb.0c09081. Epub 2020 Dec 8. J Phys Chem B. 2020. PMID: 33289567 Free PMC article.
-
Structural characterization of styrene-maleic acid copolymer-lipid nanoparticles (SMALPs) using EPR spectroscopy.Chem Phys Lipids. 2019 May;220:6-13. doi: 10.1016/j.chemphyslip.2019.02.003. Epub 2019 Feb 20. Chem Phys Lipids. 2019. PMID: 30796886 Free PMC article.
References
-
- Cornish V. W., Benson D. R., Altenbach C. A., Hideg K., Hubbell W. L., Schultz P. G. Site-specific incorporation of biophysical probes into proteins. Proceedings of the National Acadamy of Sciences of the United States of America. 1994;91(8):2910–2914. doi: 10.1073/pnas.91.8.2910. - DOI - PMC - PubMed
-
- Klare J. P. A Versatile Tool to Study Protein-Protein Interaction. InTech; 2012. Site-directed spin labelingand electron paramagnetic resonance (EPR) spectroscopy: - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

