DP1 receptor signaling prevents the onset of intrinsic apoptosis in eosinophils and functions as a transcriptional modulator
- PMID: 29607536
- PMCID: PMC6032830
- DOI: 10.1002/JLB.3MA1017-404R
DP1 receptor signaling prevents the onset of intrinsic apoptosis in eosinophils and functions as a transcriptional modulator
Abstract
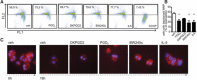
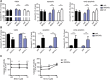
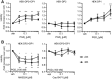
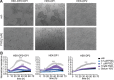
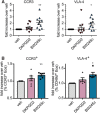
Prostaglandin (PG) D2 is the ligand for the G-protein coupled receptors DP1 (D-type prostanoid receptor 1) and DP2 (also known as chemoattractant receptor homologous molecule, expressed on Th2 cells; CRTH2). Both, DP1 and DP2 are expressed on the cellular surface of eosinophils; although it has become quite clear that PGD2 induces eosinophil migration mainly via DP2 receptors, the role of DP1 in eosinophil responses has remained elusive. In this study, we addressed how DP1 receptor signaling complements the pro-inflammatory effects of DP2. We found that PGD2 prolongs the survival of eosinophils via a DP1 receptor-mediated mechanism that inhibits the onset of the intrinsic apoptotic cascade. The DP1 agonist BW245c prevented the activation of effector caspases in eosinophils and protected mitochondrial membranes from depolarization which-as a consequence-sustained viability of eosinophils. DP1 activation in eosinophils enhanced the expression of the anti-apoptotic gene BCL-XL , but also induced pro-inflammatory genes, such as VLA-4 and CCR3. In HEK293 cells that overexpress recombinant DP1 and/or DP2 receptors, activation of DP1, but not DP2, delayed cell death and stimulated proliferation, along with induction of serum response element (SRE), a regulator of anti-apoptotic, early-response genes. We conclude that DP1 receptors promote the survival via SRE induction and induction of pro-inflammatory genes. Therefore, targeting DP1 receptors, along with DP2, may contribute to anti-inflammatory therapy in eosinophilic diseases.
Keywords: allergy; apoptosis; inflammation; prostanoids.
©2018 The Authors. Society for Leukocyte Biology Published by Wiley Periodicals, Inc.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

