Modern Approaches for Asymmetric Construction of Carbon-Fluorine Quaternary Stereogenic Centers: Synthetic Challenges and Pharmaceutical Needs
- PMID: 29608052
- PMCID: PMC6497456
- DOI: 10.1021/acs.chemrev.7b00778
Modern Approaches for Asymmetric Construction of Carbon-Fluorine Quaternary Stereogenic Centers: Synthetic Challenges and Pharmaceutical Needs
Abstract
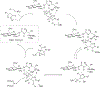
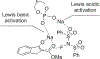
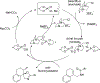
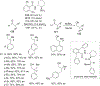
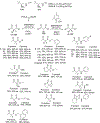
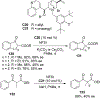
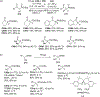
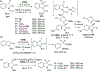
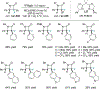
New methods for preparation of tailor-made fluorine-containing compounds are in extremely high demand in nearly every sector of chemical industry. The asymmetric construction of quaternary C-F stereogenic centers is the most synthetically challenging and, consequently, the least developed area of research. As a reflection of this apparent methodological deficit, pharmaceutical drugs featuring C-F stereogenic centers constitute less than 1% of all fluorine-containing medicines currently on the market or in clinical development. Here we provide a comprehensive review of current research activity in this area, including such general directions as asymmetric electrophilic fluorination via organocatalytic and transition-metal catalyzed reactions, asymmetric elaboration of fluorine-containing substrates via alkylations, Mannich, Michael, and aldol additions, cross-coupling reactions, and biocatalytic approaches.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



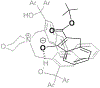
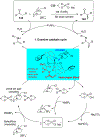
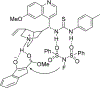
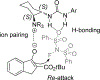

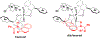








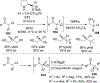
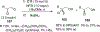
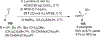


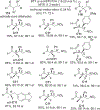


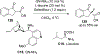


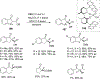
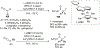
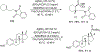
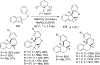

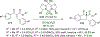
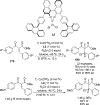
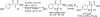

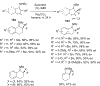
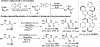
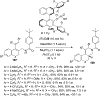
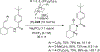
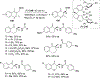


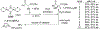


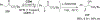

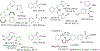
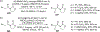
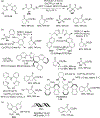
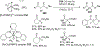


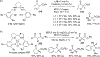
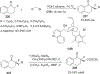
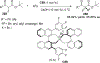
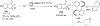
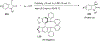
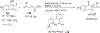

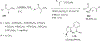
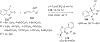
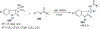
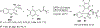
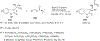
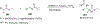
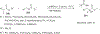
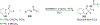
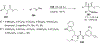
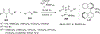


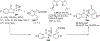
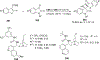

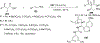
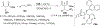


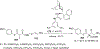
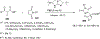
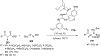
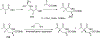
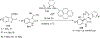
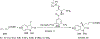

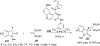
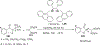
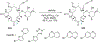
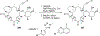
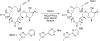
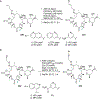


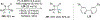

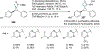
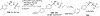
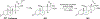
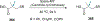
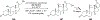
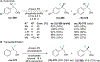



References
-
- Cameron AGW Abundances of the Elements in the Solar System. Space Sci. Rev 1973, 15, 121–146.
-
- O’Hagan D; Harper DB Fluorine-Containing Natural Products. J. Fluorine Chem 1999, 100, 127–133.
-
- Andrews SM; Cooke JA; Johnson MS Distribution of Trace Element Pollutants in a Contaminated Ecosystem Established on Metalliferous Fluorspar Tailings. 3: Fluoride. Environ. Pollut 1989, 60, 165–179. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

