Asymmetric Cu-Catalyzed 1,4-Dearomatization of Pyridines and Pyridazines without Preactivation of the Heterocycle or Nucleophile
- PMID: 29609461
- PMCID: PMC5929106
- DOI: 10.1021/jacs.8b02568
Asymmetric Cu-Catalyzed 1,4-Dearomatization of Pyridines and Pyridazines without Preactivation of the Heterocycle or Nucleophile
Abstract
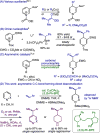
We show that a chiral copper hydride (CuH) complex catalyzes C-C bond-forming dearomatization of pyridines and pyridazines at room temperature. The catalytic reaction operates directly on free heterocycles and generates the nucleophiles in situ, eliminating the need for stoichiometric preactivation of either reaction partner; further, it is one of very few methods available for the enantioselective 1,4-dearomatization of heteroarenes. Combining the dearomatization with facile derivatization steps enables one-pot syntheses of enantioenriched pyridines and piperidines.
Conflict of interest statement
Notes
No competing financial interests have been declared
Figures
References
-
- Vitaku E, Smith DT, Njardson JT. J Med Chem. 2014;57:10257–10274. - PubMed
-
-
For reviews on pyridine dearomatization and dihydropyridine chemistry, see:
- Lavilla R. J Chem Soc, Perkin Trans. 2002;1:1141–1156.
- Ahamed M, Todd MH. Eur J Org Chem. 2010;2010:5935–5942.
- Bull JA, Mousseau JJ, Pelletier G, Charette AB. Chem Rev. 2012;112:2642–2713. - PubMed
- Zhuo CX, Zhang W, You SL. Angew Chem, Int Ed. 2012;51:12662–12686. - PubMed
- Ding Q, Zhou X, Fan R. Org Biomol Chem. 2014;12:4807–4815. - PubMed
-
-
-
For examples of 1,4-dearomatizations using copper, see:
- Piers E, Soucy M. Can J Chem. 1974;52:3563–3564.
- Comins DL, Abdullah AH. J Org Chem. 1982;47:4315–4319.
- Akiba KY, Iseki Y, Wada M. Bull Chem Soc Jpn. 1984;57:1994–1999.
-
-
-
For examples of asymmetric catalysis using activated substrates, see:
- Sun Z, Yu S, Ding Z, Ma D. J Am Chem Soc. 2007;129:9300–9301. - PubMed
- Black DA, Beveridge RE, Arndtsen BA. J Org Chem. 2008;73:1906–1910. - PubMed
- Fernández-Ibáñez MA, Maciá B, Pizzuti MG, Minnaard AJ, Feringa BL. Angew Chem, Int Ed. 2009;48:9339–9341. - PubMed
- Lutz JP, Chau ST, Doyle AG. Chem Sci. 2016;7:4105–4109. - PMC - PubMed
-
-
- Mohiti M, Rampalakos C, Feeney K, Leonori D, Aggarwal VK. Chem Sci. 2014;5:602–607.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources