Extended-release oral treprostinil in the management of pulmonary arterial hypertension: clinical evidence and experience
- PMID: 29609511
- PMCID: PMC5941659
- DOI: 10.1177/1753466618766490
Extended-release oral treprostinil in the management of pulmonary arterial hypertension: clinical evidence and experience
Abstract
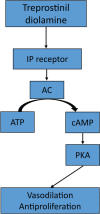
Treprostinil diolamine is the first oral prostacyclin approved for the treatment of pulmonary arterial hypertension (PAH) to improve exercise capacity. Clinical studies have demonstrated modest benefit as monotherapy, whereas no difference in exercise capacity was observed with combination therapy. However, these trials were limited by subtherapeutic dosing owing to intolerable adverse effects. Prostacyclin-related adverse effects, such as nausea, diarrhea, headache, flushing, and jaw pain, are prevalent. More recent pharmacokinetic and clinical studies illustrate the dose-response relationship and the importance of achieving clinically effective doses. Therefore, efforts to improve tolerability are paramount. Oral treprostinil is recommended to be administered three times daily in order to facilitate more rapid titration, higher doses achieved, and improved tolerability. Oral treprostinil has also been studied in carefully selected, stable patients that transitioned from parenteral or inhaled therapy with close monitoring for late deterioration. Ongoing clinical trials will determine the long-term effects of higher doses of oral treprostinil on clinical outcomes. This review describes the clinical evidence and practical experience with the use of oral treprostinil for PAH.
Keywords: oral treprostinil; prostacyclins; pulmonary arterial hypertension; pulmonary hypertension; treprostinil diolamine.
Conflict of interest statement
Figures
Similar articles
-
Rapid transition from inhaled iloprost to inhaled treprostinil in patients with pulmonary arterial hypertension.Cardiovasc Ther. 2013 Feb;31(1):38-44. doi: 10.1111/1755-5922.12008. Cardiovasc Ther. 2013. PMID: 22970909 Free PMC article. Clinical Trial.
-
Inhaled treprostinil for the treatment of pulmonary arterial hypertension.Expert Rev Respir Med. 2012 Jun;6(3):255-65. doi: 10.1586/ers.12.23. Expert Rev Respir Med. 2012. PMID: 22788940 Review.
-
Oral treprostinil for the treatment of pulmonary arterial hypertension in patients receiving background endothelin receptor antagonist and phosphodiesterase type 5 inhibitor therapy (the FREEDOM-C2 study): a randomized controlled trial.Chest. 2013 Sep;144(3):952-958. doi: 10.1378/chest.12-2875. Chest. 2013. PMID: 23669822 Clinical Trial.
-
Treprostinil for the treatment of pulmonary arterial hypertension.Expert Rev Cardiovasc Ther. 2013 Jan;11(1):13-25. doi: 10.1586/erc.12.160. Expert Rev Cardiovasc Ther. 2013. PMID: 23259441 Review.
-
Efficacy and safety of oral treprostinil monotherapy for the treatment of pulmonary arterial hypertension: a randomized, controlled trial.Circulation. 2013 Feb 5;127(5):624-33. doi: 10.1161/CIRCULATIONAHA.112.124388. Epub 2013 Jan 10. Circulation. 2013. PMID: 23307827 Clinical Trial.
Cited by
-
Management of Pulmonary Arterial Hypertension in the Pediatric Patient.Curr Cardiol Rep. 2019 Nov 28;21(12):162. doi: 10.1007/s11886-019-1229-2. Curr Cardiol Rep. 2019. PMID: 31781972 Free PMC article. Review.
-
Pharmacogenomics in the Management of Pulmonary Arterial Hypertension: Current Perspectives.Pharmgenomics Pers Med. 2023 Jul 11;16:729-737. doi: 10.2147/PGPM.S361222. eCollection 2023. Pharmgenomics Pers Med. 2023. PMID: 37457231 Free PMC article. Review.
-
A pilot study of oral treprostinil pharmacogenomics and treatment persistence in patients with pulmonary arterial hypertension.Ther Adv Respir Dis. 2021 Jan-Dec;15:17534666211013688. doi: 10.1177/17534666211013688. Ther Adv Respir Dis. 2021. PMID: 33929912 Free PMC article.
-
Comparative effectiveness of oral prostacyclin pathway drugs on hospitalization in patients with pulmonary hypertension in the United States: a retrospective database analysis.Pulm Circ. 2020 Nov 10;10(4):2045894020911831. doi: 10.1177/2045894020911831. eCollection 2020 Oct-Dec. Pulm Circ. 2020. PMID: 33240480 Free PMC article.
References
-
- Humbert M, Sitbon O, Chaouat A, et al. Survival in patients with idiopathic, familial, and anorexigen-associated pulmonary arterial hypertension in the modern management era. Circulation 2010; 122: 156–163. - PubMed
-
- Waxman AB. Oral prostacyclin therapy for pulmonary arterial hypertension: another step forward. Circulation 2013; 127: 563–565. - PubMed
-
- Jing ZC, Parikh K, Pulido T, et al. Efficacy and safety of oral treprostinil monotherapy for the treatment of pulmonary arterial hypertension: a randomized, controlled trial. Circulation 2013; 127: 624–633. - PubMed
-
- Tapson VF, Torres F, Kermeen F, et al. Oral treprostinil for the treatment of pulmonary arterial hypertension in patients on background endothelin receptor antagonist and/or phosphodiesterase type 5 inhibitor therapy (the FREEDOM-C study): a randomized controlled trial. Chest 2012; 142: 1383–1390. - PubMed
-
- Tapson VF, Jing ZC, Xu KF, et al. Oral treprostinil for the treatment of pulmonary arterial hypertension in patients receiving background endothelin receptor antagonist and phosphodiesterase type 5 inhibitor therapy (the FREEDOM-C2 study): a randomized controlled trial. Chest 2013; 144: 952–958. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases