Ginger (Zingiber officinale) phytochemicals-gingerenone-A and shogaol inhibit SaHPPK: molecular docking, molecular dynamics simulations and in vitro approaches
- PMID: 29609660
- PMCID: PMC5879566
- DOI: 10.1186/s12941-018-0266-9
Ginger (Zingiber officinale) phytochemicals-gingerenone-A and shogaol inhibit SaHPPK: molecular docking, molecular dynamics simulations and in vitro approaches
Abstract
Background: Antibiotic resistance is a defense mechanism, harbored by pathogens to survive under unfavorable conditions. Among several antibiotic resistant microbial consortium, Staphylococcus aureus is one of the most havoc microorganisms. Staphylococcus aureus encodes a unique enzyme 6-hydroxymethyl-7,8-dihydropterin pyrophosphokinase (SaHPPK), against which, none of existing antibiotics have been reported.
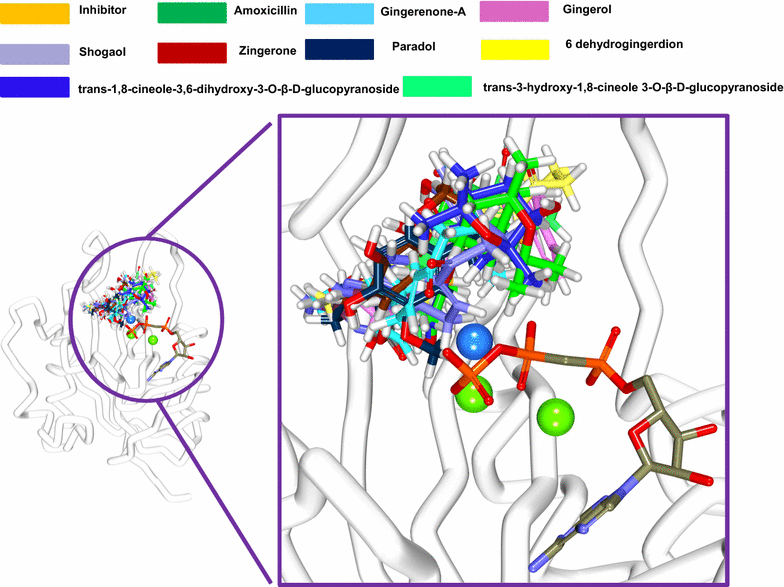
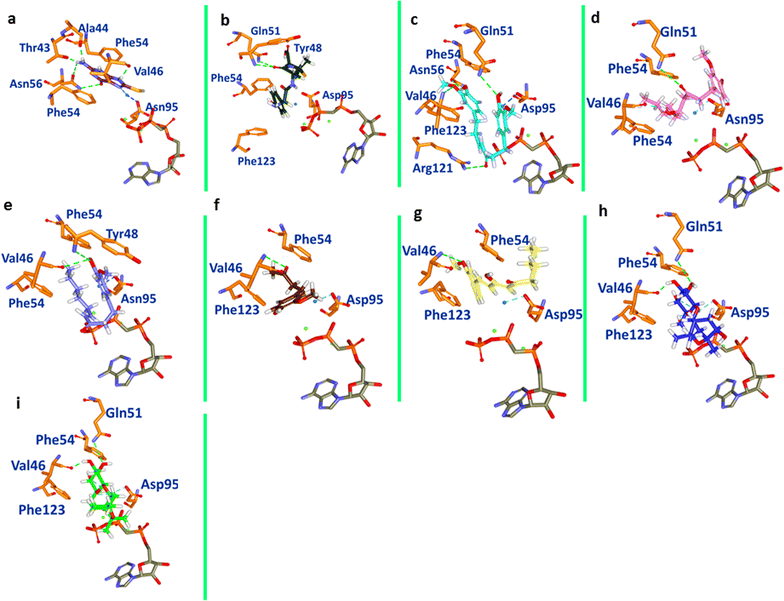
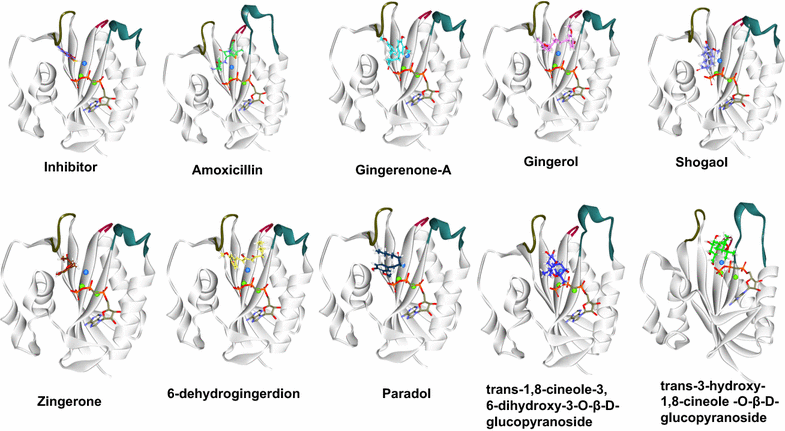
Methods: Computational approaches have been instrumental in designing and discovering new drugs for several diseases. The present study highlights the impact of ginger phytochemicals on Staphylococcus aureus SaHPPK. Herein, we have retrieved eight ginger phytochemicals from published literature and investigated their inhibitory interactions with SaHPPK. To authenticate our work, the investigation proceeds considering the known antibiotics alongside the phytochemicals. Molecular docking was performed employing GOLD and CDOCKER. The compounds with the highest dock score from both the docking programmes were tested for their inhibitory capability in vitro. The binding conformations that were seated within the binding pocket showing strong interactions with the active sites residues rendered by highest dock score were forwarded towards the molecular dynamic (MD) simulation analysis.
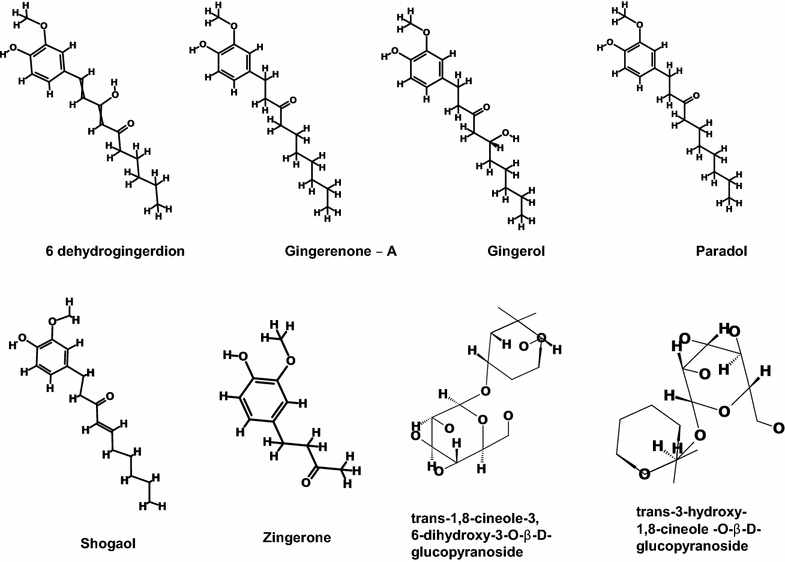
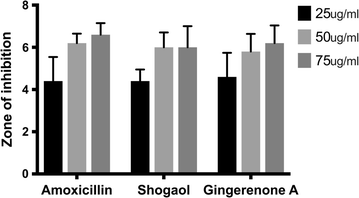
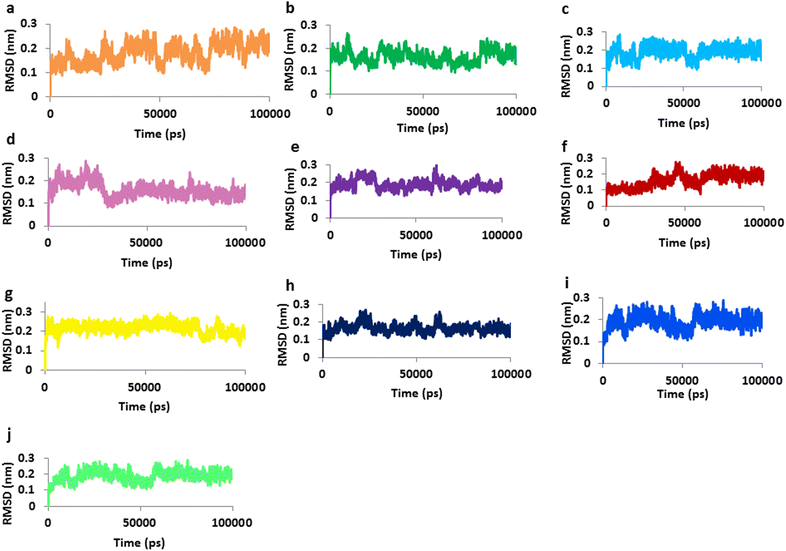
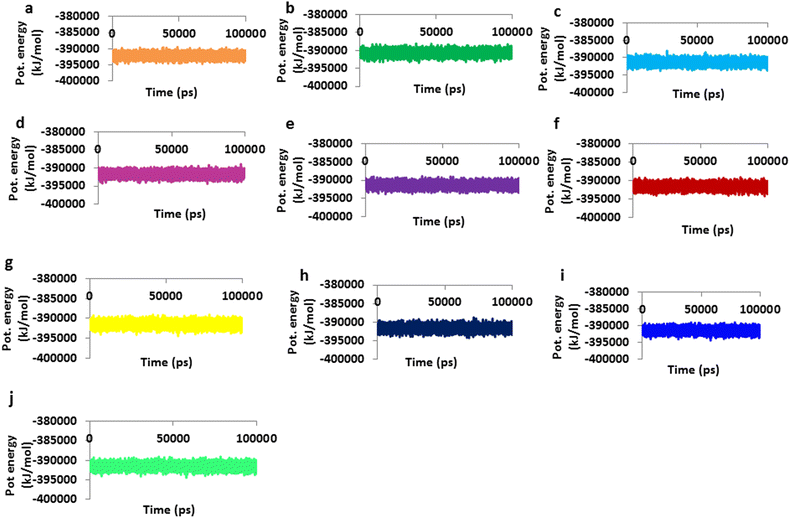
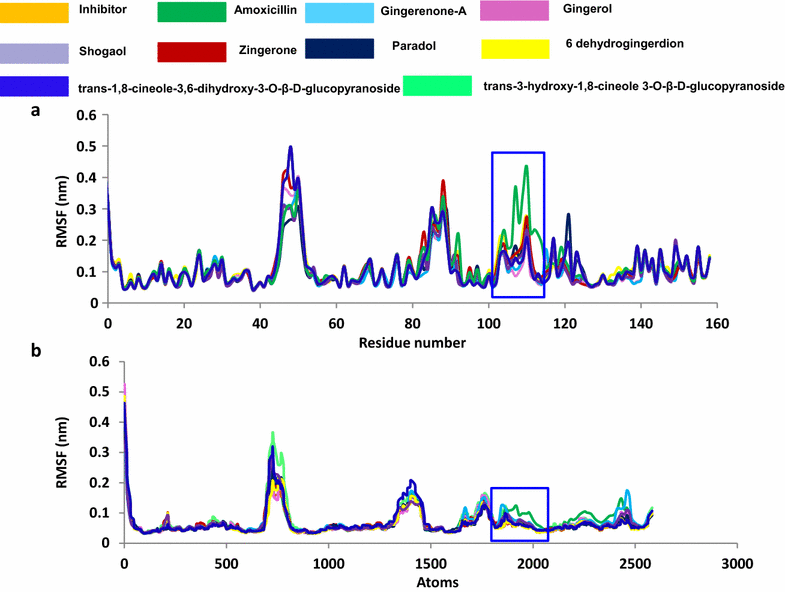
Results: Based on molecular dock scores, molecular interaction with catalytic active residues and MD simulations studies, two ginger phytochemicals, gingerenone-A and shogaol have been proposed as candidate inhibitors against Staphylococcus aureus. They have demonstrated higher dock scores than the known antibiotics and have represented interactions with the key residues within the active site. Furthermore, these compounds have rendered considerable inhibitory activity when tested in vitro. Additionally, their superiority was corroborated by stable MD results conducted for 100 ns employing GROMACS package.
Conclusions: Finally, we suggest that gingerenone-A and shogaol may either be potential SaHPPK inhibitors or can be used as fundamental platforms for novel SaHPPK inhibitor development.
Keywords: 6-Hydroxymethyl-7,8-dihydropterin pyrophosphokinase; GOLD; Ginger phytochemicals; Gingerenone-A; MD simulations; Shogaol.
Figures
Similar articles
-
Molecular docking, molecular dynamics simulation, and MM/PBSA analysis of ginger phytocompounds as a potential inhibitor of AcrB for treating multidrug-resistant Klebsiella pneumoniae infections.J Biomol Struct Dyn. 2025 Apr;43(7):3585-3601. doi: 10.1080/07391102.2023.2299741. Epub 2024 Jan 2. J Biomol Struct Dyn. 2025. PMID: 38165647
-
Pharmacotherapeutics and Molecular Mechanism of Phytochemicals in Alleviating Hormone-Responsive Breast Cancer.Oxid Med Cell Longev. 2019 Apr 4;2019:5189490. doi: 10.1155/2019/5189490. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31089409 Free PMC article.
-
Identification of Novel Aldose Reductase Inhibitors from Spices: A Molecular Docking and Simulation Study.PLoS One. 2015 Sep 18;10(9):e0138186. doi: 10.1371/journal.pone.0138186. eCollection 2015. PLoS One. 2015. PMID: 26384019 Free PMC article.
-
Cancer Chemoprevention Effects of Ginger and its Active Constituents: Potential for New Drug Discovery.Am J Chin Med. 2015;43(7):1351-63. doi: 10.1142/S0192415X15500767. Epub 2015 Oct 18. Am J Chin Med. 2015. PMID: 26477795 Review.
-
An overview of 6-shogaol: new insights into its pharmacological properties and potential therapeutic activities.Food Funct. 2024 Jul 15;15(14):7252-7270. doi: 10.1039/d3fo04753a. Food Funct. 2024. PMID: 38287779 Review.
Cited by
-
Current Understanding of the Molecular Basis of Spices for the Development of Potential Antimicrobial Medicine.Antibiotics (Basel). 2023 Jan 29;12(2):270. doi: 10.3390/antibiotics12020270. Antibiotics (Basel). 2023. PMID: 36830181 Free PMC article. Review.
-
Evaluation of tea (Camellia sinensis L.) phytochemicals as multi-disease modulators, a multidimensional in silico strategy with the combinations of network pharmacology, pharmacophore analysis, statistics and molecular docking.Mol Divers. 2023 Feb;27(1):487-509. doi: 10.1007/s11030-022-10437-1. Epub 2022 May 10. Mol Divers. 2023. PMID: 35536529 Free PMC article.
-
Unveiling Pharmacological Promise of Mangifera indica (Haribhanga) Peel Extract: Exploring an Untapped Cultivar Through Biochemical and Computational Approaches.Scientifica (Cairo). 2025 Mar 10;2025:6516268. doi: 10.1155/sci5/6516268. eCollection 2025. Scientifica (Cairo). 2025. PMID: 40225279 Free PMC article.
-
Ulcerative colitis: molecular insights and intervention therapy.Mol Biomed. 2024 Oct 10;5(1):42. doi: 10.1186/s43556-024-00207-w. Mol Biomed. 2024. PMID: 39384730 Free PMC article. Review.
-
Discovery of Potential Plant-Derived Peptide Deformylase (PDF) Inhibitors for Multidrug-Resistant Bacteria Using Computational Studies.J Clin Med. 2018 Dec 17;7(12):563. doi: 10.3390/jcm7120563. J Clin Med. 2018. PMID: 30563019 Free PMC article.
References
-
- Le Loir Y, Baron F, Gautier M. Staphylococcus aureus and food poisoning. Genet Mol Res. 2003;2:63–76. - PubMed
MeSH terms
Substances
Grants and funding
- NRF-2015M3C1A3023028/Pioneer Research Center Program through the National Research Foundation (NRF) funded by the Ministry of Science, ICT and Future Planning
- PJ01106202/Next-Generation BioGreen 21 Program
- No.10038744/Ministry of Trade, Industry & Energy(MOTIE, Korea) under Industrial Technology Innovation Program
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

