The Roles of Aquaporins in Plant Stress Responses
- PMID: 29615577
- PMCID: PMC5831814
- DOI: 10.3390/jdb4010009
The Roles of Aquaporins in Plant Stress Responses
Abstract
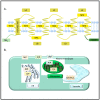
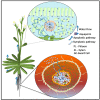
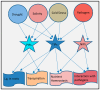
Aquaporins are membrane channel proteins ubiquitously present in all kingdoms of life. Although aquaporins were originally discovered as water channels, their roles in the transport of small neutral solutes, gasses, and metal ions are now well established. Plants contain the largest number and greatest diversity of aquaporin homologs with diverse subcellular localization patterns, gating properties, and solute specificity. The roles of aquaporins in physiological functions throughout plant growth and development are well known. As an integral regulator of plant-water relations, they are presumed to play an important role in plant defense responses against biotic and abiotic stressors. This review highlights involvement of various aquaporin homologs in plant stress responses against a variety of environmental stresses that disturb plant cell osmotic balance and nutrient homeostasis.
Keywords: abiotic stress; aquaporins; biotic stress; cold stress; drought; nutrient homeostasis; osmotic stress; salinity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Denker B.M., Smith B.L., Kuhajda F.P., Agre P. Identification, purification, and partial characterization of a novel MR 28,000 integral membrane protein from erythrocytes and renal tubules. J. Biol. Chem. 1988;263:15634–15642. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

