17-β Estradiol regulates proglucagon-derived peptide secretion in mouse and human α- and L cells
- PMID: 29618657
- PMCID: PMC5928861
- DOI: 10.1172/jci.insight.98569
17-β Estradiol regulates proglucagon-derived peptide secretion in mouse and human α- and L cells
Abstract
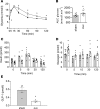
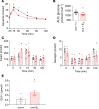
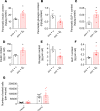
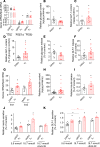
Clinical and experimental data indicate a beneficial effect of estrogens on energy and glucose homeostasis associated with improved insulin sensitivity and positive effects on insulin secretion. The aim of the study was to investigate the impact of estrogens on proglucagon-producing cells, pancreatic α cells, and enteroendocrine L cells. The consequences of sexual hormone deprivation were evaluated in ovariectomized mice (ovx). Ovx mice exhibited impaired glucose tolerance during oral glucose tolerance tests (OGTT), which was associated with decreased GLP-1 intestinal and pancreatic secretion and content, an effect that was reversed by estradiol (E2) treatment. Indeed, E2 increased oral glucose-induced GLP-1 secretion in vivo and GLP-1 secretion from primary culture of mouse and human α cells through the activation of all 3 estrogen receptors (ERs), whereas E2-induced GLP-1 secretion from mouse and human intestinal explants occurred only by ERβ activation. Underlying the implication of ERβ, its selective agonist WAY20070 was able to restore glucose tolerance in ovx mice at least partly through plasma GLP-1 increase. We conclude that E2 directly controls both α- and L cells to increase GLP-1 secretion, in addition to its effects on insulin and glucagon secretion, highlighting the potential beneficial role of the estrogenic pathway and, more particularly, of ERβ agonists to prevent type 2 diabetes.
Keywords: Diabetes; Endocrinology; Homeostasis; Metabolism; Sex hormones.
Conflict of interest statement
Figures
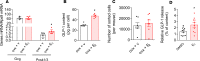




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

