MORC2, a novel oncogene, is upregulated in liver cancer and contributes to proliferation, metastasis and chemoresistance
- PMID: 29620211
- PMCID: PMC5958890
- DOI: 10.3892/ijo.2018.4333
MORC2, a novel oncogene, is upregulated in liver cancer and contributes to proliferation, metastasis and chemoresistance
Abstract
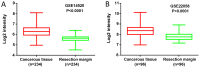
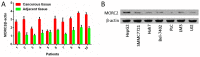
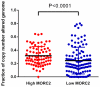
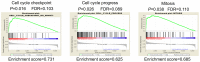
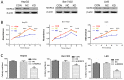
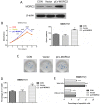
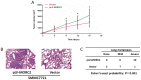
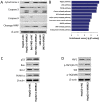
Microrchidia 2 (MORC2) is important in DNA damage repair and lipogenesis, however, the clinical and functional role of MORC2 in liver cancer remains to be fully elucidated. The aim the present study was to clarify the role of MORC2 in liver cancer. Expression profile analysis, immunohistochemical staining, reverse transcription-quantitative polymerase chain reaction analysis and western blot analysis were performed to evaluate the levels of MORC2 in liver cancer patient specimens and cell lines; subsequently the expression of MORC2 was suppressed or increased in liver cancer cells and the effects of MORC2 on the cancerous transformation of liver cancer cells were examined in vitro and in vivo. MORC2 was upregulated in liver cancer tissues, and the upregulation was associated with certain clinicopathologic features of patients with liver cancer. MORC2 knockdown caused marked inhibition of liver cancer cell proliferation and clonogenicity, whereas the overexpression of MORC2 substantially promoted liver cancer cell proliferation. In addition, the knockdown of MORC2 inhibited the migratory and invasive ability of liver cancer cells, whereas increased migration and invasion rates were observed in cells with ectopic expression of MORC2. In a model of nude mice, the overexpression of MORC2 promoted tumorigenicity and markedly enhanced pulmonary metastasis of liver cancer. Furthermore, MORC2 regulated apoptosis and its expression level had an effect on the sensitivity of liver cancer cells to doxorubicin, 5-fluorouracil and cisplatin. Mechanically, MORC2 modulated the mitochondrial apoptotic pathway, possibly in a p53-dependent manner, and its dysregulation also resulted in the abnormal activation of the Hippo pathway. For the first time, to the best of our knowledge, the present study confirmed that MORC2 was a novel oncogene in liver cancer. These results provide useful insight into the mechanism underlying the tumorigenesis and progression of liver cancer, and offers clues into potential novel liver cancer therapies.
Figures




Similar articles
-
Emerging roles of the chromatin remodeler MORC2 in cancer metabolism.Med Oncol. 2024 Aug 8;41(9):221. doi: 10.1007/s12032-024-02464-9. Med Oncol. 2024. PMID: 39117768 Review.
-
MORC2 promotes cell growth and metastasis in human cholangiocarcinoma and is negatively regulated by miR-186-5p.Aging (Albany NY). 2019 Jun 9;11(11):3639-3649. doi: 10.18632/aging.102003. Aging (Albany NY). 2019. PMID: 31180332 Free PMC article.
-
Aberrant high expression level of MORC2 is a common character in multiple cancers.Hum Pathol. 2018 Jun;76:58-67. doi: 10.1016/j.humpath.2018.03.011. Epub 2018 Mar 16. Hum Pathol. 2018. PMID: 29555576
-
Circular RNA circCDK13 suppresses cell proliferation, migration and invasion by modulating the JAK/STAT and PI3K/AKT pathways in liver cancer.Int J Oncol. 2018 Jul;53(1):246-256. doi: 10.3892/ijo.2018.4371. Epub 2018 Apr 12. Int J Oncol. 2018. PMID: 29658568
-
Novel Insights into the Role of Chromatin Remodeler MORC2 in Cancer.Biomolecules. 2023 Oct 15;13(10):1527. doi: 10.3390/biom13101527. Biomolecules. 2023. PMID: 37892209 Free PMC article. Review.
Cited by
-
Targeting Hippo signaling in cancer: novel perspectives and therapeutic potential.MedComm (2020). 2023 Oct 3;4(5):e375. doi: 10.1002/mco2.375. eCollection 2023 Oct. MedComm (2020). 2023. PMID: 37799806 Free PMC article. Review.
-
MORC2 Interactome: Its Involvement in Metabolism and Cancer.Biophys Rev. 2021 Jun 4;13(4):507-514. doi: 10.1007/s12551-021-00812-x. eCollection 2021 Aug. Biophys Rev. 2021. PMID: 34471435 Free PMC article. Review.
-
CRISPR/Cas9 system: a novel approach to overcome chemotherapy and radiotherapy resistance in cancer.Naunyn Schmiedebergs Arch Pharmacol. 2025 Apr;398(4):3373-3408. doi: 10.1007/s00210-024-03480-2. Epub 2024 Nov 19. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39560750 Review.
-
Emerging roles of the chromatin remodeler MORC2 in cancer metabolism.Med Oncol. 2024 Aug 8;41(9):221. doi: 10.1007/s12032-024-02464-9. Med Oncol. 2024. PMID: 39117768 Review.
-
Inhibition of MORC2 Mediates HDAC4 to Promote Cellular Senescence through p53/p21 Signaling Axis.Molecules. 2022 Sep 22;27(19):6247. doi: 10.3390/molecules27196247. Molecules. 2022. PMID: 36234785 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

