Tofacitinib Halts Progression of Graft Dysfunction in a Rat Model of Mixed Cellular and Humoral Rejection
- PMID: 29620612
- PMCID: PMC7228626
- DOI: 10.1097/TP.0000000000002204
Tofacitinib Halts Progression of Graft Dysfunction in a Rat Model of Mixed Cellular and Humoral Rejection
Abstract
Background: The progression from acute to chronic antibody-mediated rejection in kidney transplant recipients is usually not prevented by current therapeutic options. Here, we investigated whether the use of tofacitinib (TOFA), a Janus kinase 3 inhibitor, was capable of preventing the progression of allograft dysfunction in a Fisher-to-Lewis rat model of kidney transplantation.
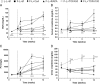
Methods: Rats were treated from the third week after transplantation to allow the development of rejection. Treatment was based on cyclosporin A, rapamycin or TOFA. Renal function was assessed at 1, 4, 8, and 12 weeks after transplantation, whereas rat survival, histological lesions, and infiltrating lymphocytes were analyzed at 12 weeks.
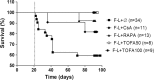
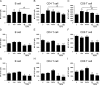
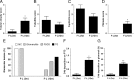
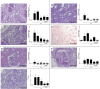
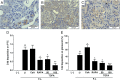
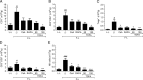
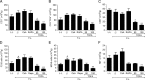
Results: Tofacitinib prolonged graft survival, preserved tubular and glomerular structures and reduced humoral damage characterized by C4d deposition. Tofacitinib was able to reduce donor-specific antibodies. In addition, T and natural killer cell graft infiltration was reduced in TOFA-treated rats. Although rapamycin-treated rats also showed prolonged graft survival, glomerular structures were more affected. Moreover, only TOFA treatment reduced the presence of T, B and natural killer cells in splenic parenchyma.
Conclusions: Tofacitinib is able to reduce the immune response generated in a rat model of kidney graft rejection, providing prolonged graft and recipient survival, better graft function, and less histological lesions.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Meier-Kriesche HU, Li S, Gruessner RW. Immunosuppression: evolution in practice and trends, 1994-2004 Am J Transplant 2006. 61111–1131 - PubMed
-
- Halloran PF. Immunosuppressive drugs for kidney transplantation N Engl J Med 2004. 3512715–2729 - PubMed
-
- Nankivell BJ, Borrows RJ, Fung CL. The natural history of chronic allograft nephropathy N Engl J Med 2003. 3492326–2333 - PubMed
-
- Dantal J, Soulillou JP. Immunosuppressive drugs and the risk of cancer after organ transplantation N Engl J Med 2005. 3521371–1373 - PubMed
-
- Terasaki PI. Humoral theory of transplantation Am J Transplant 2003. 3665–673 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

