Multifunctional biomedical imaging in physiological and pathological conditions using a NIR-II probe
- PMID: 29623009
- PMCID: PMC5879786
- DOI: 10.1002/adfm.201700995
Multifunctional biomedical imaging in physiological and pathological conditions using a NIR-II probe
Abstract
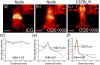
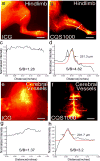
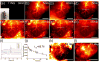
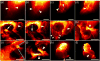
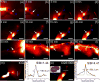
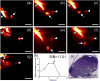
Compared with imaging in the visible (400 - 650 nm) and near-infrared window I (NIR-I, 650 - 900 nm) regions, imaging in near-infrared window II (NIR-II, 1,000-1,700 nm) is a highly promising in vivo imaging modality with improved resolution and deeper tissue penetration. In this work, a small molecule NIR-II dye,5,5'-(1H,5H-benzo[1,2-c:4,5-c'] bis[1,2,5]thiadiazole)-4,8-diyl)bis(N,N-bis(4-(3-((tert-butyldimethylsilyl)oxy)propyl)phenyl) thiophen-2-amine), has been successfully encapsulated into phospholipid vesicles to prepare a probe CQS1000. Then this novel NIR-II probe has been studied for in vivo multifunctional biological imaging. Our results indicate that the NIR-II vesicle CQS1000 can noninvasively and dynamically visualize and monitor many physiological and pathological conditions of circulatory systems, including lymphatic drainage and routing, angiogenesis of tumor and vascular deformity such as arterial thrombus formation and ischemia with high spatial and temporal resolution. More importantly, by virtue of the favorable half-life of blood circulation of CQS1000, NIR-II imaging is capable of aiding us to accomplish precise resection of tumor such as osteosarcoma, and to accelerate the process of lymph nodes dissection to complete sentinel lymph node biopsy for better decision-making during the tumor surgery. Overall, CQS1000 is a highly promising NIR-II probe for multifunctional biomedical imaging in physiological and pathological conditions, surpassing traditional NIR-I imaging modality and pathologic assessments for clinical diagnosis and treatment.
Keywords: NIR-II; fluorescence imaging; sentinel lymph node; tumor; vascular and lymphatic system.
Conflict of interest statement
Conflict of interest No conflict of interest.
Figures


References
-
- Zhu Q, DeFusco PA, Ricci A, Jr, Cronin EB, Hegde PU, Kane M, Tavakoli B, Xu Y, Hart J, Tannenbaum SH. Radiology. 2013;266:433. - PMC - PubMed
- Golijanin J, Amin A, Moshnikova A, Brito JM, Tran TY, Adochite R-C, Andreev GO, Crawford T, Engelman DM, Andreev OA. Proceedings of the National Academy of Sciences. 2016;113:11829. - PMC - PubMed
- Sun Y, Ma X, Cheng K, Wu B, Duan J, Chen H, Bu L, Zhang R, Hu X, Deng Z. Angewandte Chemie International Edition. 2015;54:5981. - PubMed
-
- Okochi O, Kaneko T, Sugimoto H, Inoue S, Takeda S, Nakao A. Journal of Surgical Research. 2002;103:109. - PubMed
-
- Meier R, Thuermel K, Noël PB, Moog P, Sievert M, Ahari C, Nasirudin RA, Golovko D, Haller B, Ganter C. Radiology. 2014;270:176. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
