Efficacy and Safety of Rapid-Acting Insulin Analogs in Special Populations with Type 1 Diabetes or Gestational Diabetes: Systematic Review and Meta-Analysis
- PMID: 29623593
- PMCID: PMC5984914
- DOI: 10.1007/s13300-018-0411-7
Efficacy and Safety of Rapid-Acting Insulin Analogs in Special Populations with Type 1 Diabetes or Gestational Diabetes: Systematic Review and Meta-Analysis
Abstract
Introduction: To assess the efficacy and safety of three available rapid-acting insulin analogs (insulins lispro, aspart and glulisine, respectively) in pregnant women, children/adolescents and people using continuous subcutaneous insulin infusion (CSII) with type 1 diabetes.
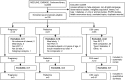
Methods: PubMed, EMBASE and Cochrane Reviews were searched electronically, and their bibliographies examined to identify suitable studies for review and inclusion in a meta-analysis. Eligible studies were randomized controlled trials that reported data on relevant clinical outcomes. A different reviewer abstracted data for each of the three subpopulations, and one reviewer abstracted data for all three. Any differences were resolved by consensus or by consulting a fourth reviewer.
Results: In people on CSII, rapid-acting insulin analogs lowered postprandial plasma glucose post-breakfast to a greater extent than did regular human insulin (RHI) (mean difference: - 1.63 mmol/L [95% confidence interval - 1.71; - 1.54]), with a comparable risk of hypoglycemia and a trend for lower glycated hemoglobin. In the pediatric population, glycemic control was similar with rapid-acting insulin analogs and RHI, with no safety concerns. Meta-analysis indicated severe hypoglycemic events were comparable for rapid-acting insulin analogs versus RHI (risk difference: 0.00 [95% confidence interval - 0.01; 0.01]). In the pregnancy group, insulin lispro and insulin aspart were safe and effective for both mother and fetus, with glycemic control being at least as good as with RHI. There were no data on insulin glulisine during pregnancy.
Conclusion: Rapid-acting insulin analogs appear generally safe and effective in these special populations; however, additional trials would be helpful.
Funding: Novo Nordisk A/S.
Keywords: CSII; Pediatrics; Pregnancy; Rapid-acting insulin analogs; Type 1 diabetes.
Figures

References
-
- American Diabetes Association Standards of medical care in diabetes—2017. Diabetes Care. 2017;40(Suppl. 1):S1–S135.
-
- Eli Lilly. Humalog prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2013/020563s115lbl.pdf; 2013. Accessed 29 Jan 2018.
-
- Novo Nordisk. NovoLog prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2015/020986s082lbl.pdf; 2015. Accessed 29 Jan 2018.
-
- Sanofi Aventis. Apidra prescribing information. www.accessdata.fda.gov/drugsatfda_docs/label/2015/021629s030lbl.pdf; 2015. Accessed 29 Jan 2018.
-
- Eli Lilly. Humalog product information. www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/000...; 2017. Accessed 29 Jan 2018.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

