Pattern separation and completion of distinct axonal inputs transmitted via micro-tunnels between co-cultured hippocampal dentate, CA3, CA1 and entorhinal cortex networks
- PMID: 29623900
- PMCID: PMC6021217
- DOI: 10.1088/1741-2552/aabc20
Pattern separation and completion of distinct axonal inputs transmitted via micro-tunnels between co-cultured hippocampal dentate, CA3, CA1 and entorhinal cortex networks
Abstract
Objective: Functions ascribed to the hippocampal sub-regions for encoding episodic memories include the separation of activity patterns propagated from the entorhinal cortex (EC) into the dentate gyrus (DG) and pattern completion in CA3 region. Since a direct assessment of these functions is lacking at the level of specific axonal inputs, our goal is to directly measure the separation and completion of distinct axonal inputs in engineered pairs of hippocampal sub-regional circuits.
Approach: We co-cultured EC-DG, DG-CA3, CA3-CA1 or CA1-EC neurons in a two-chamber PDMS device over a micro-electrode array (MEA60), inter-connected via distinct axons that grow through the micro-tunnels between the compartments. Taking advantage of the axonal accessibility, we quantified pattern separation and completion of the evoked activity transmitted through the tunnels from source into target well. Since pattern separation can be inferred when inputs are more correlated than outputs, we first compared the correlations among axonal inputs with those of target somata outputs. We then compared, in an analog approach, the distributions of correlation distances between rate patterns of the axonal inputs inside the tunnels with those of the somata outputs evoked in the target well. Finally, in a digital approach, we measured the spatial population distances between binary patterns of the same axonal inputs and somata outputs.
Main results: We found the strongest separation of the propagated axonal inputs when EC was axonally connected to DG, with a decline in separation to CA3 and to CA1 for both rate and digital approaches. Furthermore, the digital approach showed stronger pattern completion in CA3, then CA1 and EC.
Significance: To the best of our knowledge, these are the first direct measures of pattern separation and completion for axonal transmission to the somata target outputs at the rate and digital population levels in each of four stages of the EC-DG-CA3-CA1 circuit.
Figures





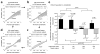

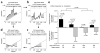

Similar articles
-
Hippocampal network axons respond to patterned theta burst stimulation with lower activity of initially higher spike train similarity from EC to DG and later similarity of axons from CA1 to EC.J Neural Eng. 2023 Sep 12;20(5). doi: 10.1088/1741-2552/acf68a. J Neural Eng. 2023. PMID: 37666242
-
Sparse and Specific Coding during Information Transmission between Co-cultured Dentate Gyrus and CA3 Hippocampal Networks.Front Neural Circuits. 2017 Mar 6;11:13. doi: 10.3389/fncir.2017.00013. eCollection 2017. Front Neural Circuits. 2017. PMID: 28321182 Free PMC article.
-
Repeating Spatial-Temporal Motifs of CA3 Activity Dependent on Engineered Inputs from Dentate Gyrus Neurons in Live Hippocampal Networks.Front Neural Circuits. 2016 Jun 28;10:45. doi: 10.3389/fncir.2016.00045. eCollection 2016. Front Neural Circuits. 2016. PMID: 27445701 Free PMC article.
-
Tracking the flow of hippocampal computation: Pattern separation, pattern completion, and attractor dynamics.Neurobiol Learn Mem. 2016 Mar;129:38-49. doi: 10.1016/j.nlm.2015.10.008. Epub 2015 Oct 26. Neurobiol Learn Mem. 2016. PMID: 26514299 Free PMC article. Review.
-
Function of local circuits in the hippocampal dentate gyrus-CA3 system.Neurosci Res. 2019 Mar;140:43-52. doi: 10.1016/j.neures.2018.11.003. Epub 2018 Nov 5. Neurosci Res. 2019. PMID: 30408501 Review.
Cited by
-
Pathway-like Activation of 3D Neuronal Constructs with an Optical Interface.Biosensors (Basel). 2025 Mar 12;15(3):179. doi: 10.3390/bios15030179. Biosensors (Basel). 2025. PMID: 40136976 Free PMC article.
-
Robust and consistent measures of pattern separation based on information theory and demonstrated in the dentate gyrus.PLoS Comput Biol. 2024 Feb 20;20(2):e1010706. doi: 10.1371/journal.pcbi.1010706. eCollection 2024 Feb. PLoS Comput Biol. 2024. PMID: 38377108 Free PMC article.
-
Recent Progress and Perspectives on Neural Chip Platforms Integrating PDMS-Based Microfluidic Devices and Microelectrode Arrays.Micromachines (Basel). 2023 Mar 23;14(4):709. doi: 10.3390/mi14040709. Micromachines (Basel). 2023. PMID: 37420942 Free PMC article. Review.
-
Opportunities and Challenges of Brain-on-a-Chip Interfaces.Cyborg Bionic Syst. 2025 Jun 17;6:0287. doi: 10.34133/cbsystems.0287. eCollection 2025. Cyborg Bionic Syst. 2025. PMID: 40530005 Free PMC article. Review.
-
Microfluidic Bi-Layer Platform to Study Functional Interaction between Co-Cultured Neural Networks with Unidirectional Synaptic Connectivity.Micromachines (Basel). 2023 Apr 11;14(4):835. doi: 10.3390/mi14040835. Micromachines (Basel). 2023. PMID: 37421068 Free PMC article.
References
-
- Andersen P, Morris R, Amaral D, Bliss T, O'Keefe J. The Hippocampus Book. 2006. - DOI
-
- Baranes D, López-García JC, Chen M, Bailey CH, Kandel ER. Reconstitution of the hippocampal mossy fiber and associational-commissural pathways in a novel dissociated cell culture system. Proceedings of the National Academy of Sciences of the United States of America. 1996;93:4706–4711. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
