Montelukast for bronchiolitis obliterans syndrome after lung transplantation: A randomized controlled trial
- PMID: 29624575
- PMCID: PMC5889063
- DOI: 10.1371/journal.pone.0193564
Montelukast for bronchiolitis obliterans syndrome after lung transplantation: A randomized controlled trial
Abstract
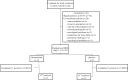
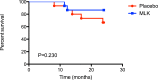
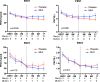
Bronchiolitis obliterans syndrome (BOS) remains the major problem which precludes long-term survival after lung transplantation. Previously, an open label pilot study from our group demonstrated a possible beneficial effect of montelukast in progressive BOS patients with low airway neutrophilia (<15%), and already on azithromycin treatment, in whom the further decline in pulmonary function was attenuated. This was, however, a non-randomized and non-placebo controlled trial. The study design is a single center, prospective, interventional, randomized, double blind, placebo-controlled trial, with a two arm parallel group design and an allocation ratio of 1:1. Randomization to additional montelukast (10 mg/day, n = 15) or placebo (n = 15) was performed from 2010 to 2014 at the University Hospitals Leuven (Leuven, Belgium) in all consecutive patients with late-onset (>2years posttransplant) BOS ≥1. Primary end-point was freedom from graft loss 1 year after randomization; secondary end-points were acute rejection, lymphocytic bronchiolitis, respiratory infection rate; and change in FEV1, airway and systemic inflammation during the study period. Graft loss at 1 y and 2y was similar in both groups (respectively p = 0. 981 and p = 0.230). Montelukast had no effect on lung function decline in the overall cohort. However, in a post-hoc subanalysis of BOS stage 1 patients, montelukast attenuated further decline of FEV1 during the study period, both in absolute (L) (p = 0.008) and % predicted value (p = 0.0180). A linear mixed model confirmed this association. Acute rejection, lymphocytic bronchiolitis, respiratory infections, systemic and airway inflammation were comparable between groups over the study period. This randomized controlled trial showed no additional survival benefit with montelukast compared to placebo, although the study was underpowered. The administration of montelukast was associated with an attenuation of the rate of FEV1 decline, however, only in recipients with late-onset BOS stage 1.
Conflict of interest statement
Figures

References
-
- Yusen RD, Edwards LB, Kucheryavaya AY, et al. The registry of the International Society for Heart and Lung Transplantation: thirty-first adult lung and heart-lung transplant report—2014; focus theme: retransplantation. J Heart Lung Transplant 2014; 33:1009–24. doi: 10.1016/j.healun.2014.08.004 - DOI - PubMed
-
- Burlingham WJ, Love RB, Jankowska-Gan E, et al. IL-17-dependent cellular immunity to collagen type V predisposes to obliterative bronchiolitis in human lung transplants. J Clin Invest 2007;117:3498–506. doi: 10.1172/JCI28031 - DOI - PMC - PubMed
-
- Sato M, Waddell TK, Wagnetz U, et al. Restrictive allograft syndrome (RAS): a novel form of chronic lung allograft dysfunction. J Heart Lung Transplant 2011;30:735–42. doi: 10.1016/j.healun.2011.01.712 - DOI - PubMed
-
- Verleden GM, Raghu G, Meyer KC, et al. A new classification system for chronic lung allograft dysfunction. J Heart Lung Transplant 2014;33:127–33. doi: 10.1016/j.healun.2013.10.022 - DOI - PubMed
-
- Christie JD, Edwards LB, Kucheryavaya, et al. The Registry of the International Society for Heart and Lung Transplantation: 29th adult lung and heart-lung transplant report-2012. J Heart Lung Transplant 2012;31:1073–86. doi: 10.1016/j.healun.2012.08.004 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

