Choroidal and Sub-Retinal Pigment Epithelium Caverns: Multimodal Imaging and Correspondence with Friedman Lipid Globules
- PMID: 29625839
- PMCID: PMC6321740
- DOI: 10.1016/j.ophtha.2018.02.036
Choroidal and Sub-Retinal Pigment Epithelium Caverns: Multimodal Imaging and Correspondence with Friedman Lipid Globules
Abstract
Purpose: To survey Friedman lipid globules by high-resolution histologic examination and to compare with multimodal imaging of hyporeflective caverns in eyes with geographic atrophy (GA) secondary to age-related macular (AMD) and other retinal diseases.
Design: Histologic survey of donor eyes with and without AMD. Clinical case series with multimodal imaging analysis.
Participants: Donor eyes (n = 139; 26 with early AMD, 13 with GA, 40 with nAMD, 52 with a healthy macula, and 8 with other or unknown characteristics) and 41 eyes of 28 participants with GA (n = 16), nAMD (n = 8), Stargardt disease (n = 4), cone dystrophy (n = 2), pachychoroid spectrum (n = 6), choroidal hemangioma (n = 1), and healthy eyes (n = 4).
Methods: Donor eyes were prepared for macula-wide epoxy resin sections through the foveal and perifoveal area. In patients, caverns were identified as nonreflective spaces on OCT images. Multimodal imaging included color and red-free fundus photography; fundus autofluorescence; fluorescein and, indocyanine green angiography; OCT angiography; near-infrared reflectance; and confocal multispectral (MultiColor [Spectralis, Heidelberg Engineering, Germany]) imaging.
Main outcome measures: Presence and morphologic features of globules, and presence and appearance of caverns on multimodal imaging.
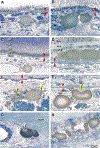
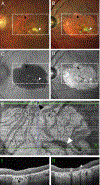
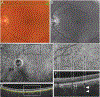
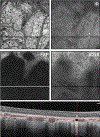
Results: Globules were found primarily in the inner choroidal stroma (91.0%), but also localized to the sclera (4.9%) and neovascular membranes (2.1%). Mean diameters of solitary and multilobular globules were 58.9±37.8 μm and 65.4±27.9 μm, respectively. Globules showed morphologic signs of dynamism including pitting, dispersion, disintegration, and crystal formation. Evidence for inflammation in the surrounding tissue was absent. En face OCT rendered sharply delimited hyporeflective areas as large as choroidal vessels, frequently grouped around choroid vessels or in the neovascular tissue. Cross-sectional OCT revealed a characteristic posterior hypertransmission. OCT angiography showed absence of flow signal within caverns.
Conclusions: Based on prior literature documenting OCT signatures of tissue lipid in atheroma and nAMD, we speculate that caverns are lipid rich. Globules, with similar sizes and tissue locations in AMD and healthy persons, are candidates for histologic correlates of caverns. The role of globules in chorioretinal physiologic features, perhaps as a lipid depot for photoreceptor metabolism, is approachable through clinical imaging.
Copyright © 2018. Published by Elsevier Inc.
Figures




Comment in
-
Re: Dolz-Marco et al.: Choroidal and sub-retinal pigment epithelium caverns: multimodal imaging and correspondence with Friedman lipid globules (Ophthalmology. 2018;125:1287-1301).Ophthalmology. 2019 Jul;126(7):e53-e54. doi: 10.1016/j.ophtha.2019.01.019. Ophthalmology. 2019. PMID: 31229014 No abstract available.
-
Reply.Ophthalmology. 2019 Jul;126(7):e54-e55. doi: 10.1016/j.ophtha.2019.01.020. Ophthalmology. 2019. PMID: 31229015 No abstract available.
References
-
- Spaide RF, Koizumi H, Pozonni MC. Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol 2008;146(4):496–500. - PubMed
-
- Tan CS, Ouyang Y, Ruiz H, Sadda SR. Diurnal variation of choroidal thickness in normal, healthy subjects measured by spectral domain optical coherence tomography. Investigative ophthalmology & visual science 2012;53(1):261–266. - PubMed
-
- Jaensch PA. Die degenerative Verfettung am Auge. III. Die krankhafte Verfettung der Gefäßhaut. Graefes Arhiv für Ophthalmologie 1935;133:517–531.
-
- Friedman E, Smith TR. Clinical and pathological study of choroidal lipid globules. Arch Ophthalmol 1966;75(3):334–336. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

