Leucine-rich repeat-containing synaptic adhesion molecules as organizers of synaptic specificity and diversity
- PMID: 29628503
- PMCID: PMC5938020
- DOI: 10.1038/s12276-017-0023-8
Leucine-rich repeat-containing synaptic adhesion molecules as organizers of synaptic specificity and diversity
Abstract
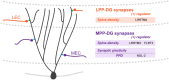
The brain harbors billions of neurons that form distinct neural circuits with exquisite specificity. Specific patterns of connectivity between distinct neuronal cell types permit the transfer and computation of information. The molecular correlates that give rise to synaptic specificity are incompletely understood. Recent studies indicate that cell-surface molecules are important determinants of cell type identity and suggest that these are essential players in the specification of synaptic connectivity. Leucine-rich repeat (LRR)-containing adhesion molecules in particular have emerged as key organizers of excitatory and inhibitory synapses. Here, we discuss emerging evidence that LRR proteins regulate the assembly of specific connectivity patterns across neural circuits, and contribute to the diverse structural and functional properties of synapses, two key features that are critical for the proper formation and function of neural circuits.
Conflict of interest statement
This manuscript is commissioned by Dr. Eunjoon Kim and Jaewon Ko for a 2018 special issue on synapse assembly, neural circuit development, and brain disorders.
Figures


Similar articles
-
Control of neural circuit formation by leucine-rich repeat proteins.Trends Neurosci. 2014 Oct;37(10):539-50. doi: 10.1016/j.tins.2014.07.004. Epub 2014 Aug 14. Trends Neurosci. 2014. PMID: 25131359 Free PMC article. Review.
-
The leucine-rich repeat superfamily of synaptic adhesion molecules: LRRTMs and Slitrks.Mol Cells. 2012 Oct;34(4):335-40. doi: 10.1007/s10059-012-0113-3. Epub 2012 Jul 4. Mol Cells. 2012. PMID: 22767246 Free PMC article. Review.
-
The NGL family of leucine-rich repeat-containing synaptic adhesion molecules.Mol Cell Neurosci. 2009 Sep;42(1):1-10. doi: 10.1016/j.mcn.2009.05.008. Epub 2009 May 23. Mol Cell Neurosci. 2009. PMID: 19467332 Review.
-
Molecular mechanisms of synaptic specificity in developing neural circuits.Neuron. 2010 Oct 6;68(1):9-18. doi: 10.1016/j.neuron.2010.09.007. Neuron. 2010. PMID: 20920787 Free PMC article. Review.
-
SynCAMs extend their functions beyond the synapse.Eur J Neurosci. 2014 Jun;39(11):1752-60. doi: 10.1111/ejn.12544. Epub 2014 Mar 15. Eur J Neurosci. 2014. PMID: 24628990 Review.
Cited by
-
LRRTM3 Genetic Variations, rs1925575, and rs1925608 Contributed to Autism Spectrum Disorder Trait Severity: An Observation in The Indian Probands.Int J Mol Cell Med. 2023;12(4):350-360. doi: 10.22088/IJMCM.BUMS.12.4.350. Int J Mol Cell Med. 2023. PMID: 39006200 Free PMC article.
-
LRIT3 expression in cone photoreceptors restores post-synaptic bipolar cell signalplex assembly and partial function in Lrit3 -/- mice.iScience. 2023 Mar 24;26(4):106499. doi: 10.1016/j.isci.2023.106499. eCollection 2023 Apr 21. iScience. 2023. PMID: 37091241 Free PMC article.
-
Insight into the Association between Slitrk Protein and Neurodevelopmental and Neuropsychiatric Conditions.Biomolecules. 2024 Aug 26;14(9):1060. doi: 10.3390/biom14091060. Biomolecules. 2024. PMID: 39334827 Free PMC article. Review.
-
Synaptic recognition molecules in development and disease.Curr Top Dev Biol. 2021;142:319-370. doi: 10.1016/bs.ctdb.2020.12.009. Epub 2021 Feb 12. Curr Top Dev Biol. 2021. PMID: 33706921 Free PMC article.
-
Synaptic signatures and disease vulnerabilities of layer 5 pyramidal neurons.Nat Commun. 2025 Jan 2;16(1):228. doi: 10.1038/s41467-024-55470-w. Nat Commun. 2025. PMID: 39747884 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

