Dual vicinal functionalisation of heterocycles via an interrupted Pummerer coupling/[3,3]-sigmatropic rearrangement cascade
- PMID: 29629145
- PMCID: PMC5870476
- DOI: 10.1039/c7sc04723a
Dual vicinal functionalisation of heterocycles via an interrupted Pummerer coupling/[3,3]-sigmatropic rearrangement cascade
Abstract
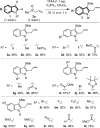
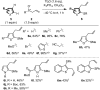
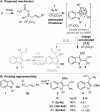
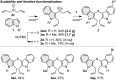
A dual vicinal functionalisation cascade involving the union of heterocycles and allyl sulfoxides is described. In particular, the approach provides efficient one-step access to biologically relevant and synthetically important C3 thio, C2 carbo substituted indoles. The reaction operates under mild, metal free conditions and without directing groups, via an interrupted Pummerer coupling of activated allyl sulfoxides, generating allyl heteroaryl sulfonium salts that are predisposed to a charge accelerated [3,3]-sigmatropic rearrangement.
Figures


Similar articles
-
Synthesis of C2 Substituted Benzothiophenes via an Interrupted Pummerer/[3,3]-Sigmatropic/1,2-Migration Cascade of Benzothiophene S-Oxides.Angew Chem Int Ed Engl. 2018 May 14;57(20):5759-5764. doi: 10.1002/anie.201801982. Epub 2018 Apr 10. Angew Chem Int Ed Engl. 2018. PMID: 29528177
-
Cascades of Interrupted Pummerer Reaction-Sigmatropic Rearrangement.Chem Rec. 2017 Nov;17(11):1156-1167. doi: 10.1002/tcr.201700017. Epub 2017 May 10. Chem Rec. 2017. PMID: 28488753 Review.
-
An Interrupted Pummerer/Nickel-Catalysed Cross-Coupling Sequence.Angew Chem Int Ed Engl. 2018 Jul 26;57(31):9785-9789. doi: 10.1002/anie.201805396. Epub 2018 Jul 5. Angew Chem Int Ed Engl. 2018. PMID: 29882623
-
Sulfonium-aided coupling of aromatic rings via sigmatropic rearrangement.Proc Jpn Acad Ser B Phys Biol Sci. 2022;98(4):190-205. doi: 10.2183/pjab.98.012. Proc Jpn Acad Ser B Phys Biol Sci. 2022. PMID: 35400695 Free PMC article.
-
[Some recent advances in Pummerer-type reactions].Yakugaku Zasshi. 1997 May;117(5):282-97. doi: 10.1248/yakushi1947.117.5_282. Yakugaku Zasshi. 1997. PMID: 9194396 Review. Japanese.
Cited by
-
Bond-Forming and -Breaking Reactions at Sulfur(IV): Sulfoxides, Sulfonium Salts, Sulfur Ylides, and Sulfinate Salts.Chem Rev. 2019 Jul 24;119(14):8701-8780. doi: 10.1021/acs.chemrev.9b00111. Epub 2019 Jun 25. Chem Rev. 2019. PMID: 31243998 Free PMC article.
-
Iridium-Catalyzed Enantioselective Intermolecular Indole C2-Allylation.Angew Chem Int Ed Engl. 2020 May 4;59(19):7598-7604. doi: 10.1002/anie.202001956. Epub 2020 Mar 11. Angew Chem Int Ed Engl. 2020. PMID: 32091146 Free PMC article.
-
Diastereoselective Indole-Dearomative Cope Rearrangements by Compounding Minor Driving Forces.Org Lett. 2022 May 27;24(20):3726-3730. doi: 10.1021/acs.orglett.2c01381. Epub 2022 May 16. Org Lett. 2022. PMID: 35576941 Free PMC article.
-
Unified approach to synthesize diverse heterocyclics: a metal-free visible-light-promoted cyclization reaction to acquire sulfonylated spiro-trienones, coumarins and their derivatives.RSC Adv. 2025 Jul 8;15(29):23633-23642. doi: 10.1039/d5ra03553h. eCollection 2025 Jul 4. RSC Adv. 2025. PMID: 40630678 Free PMC article.
-
Sulfoxide-mediated oxidative cross-coupling of phenols.Chem Sci. 2020 Jan 15;11(7):2001-2005. doi: 10.1039/c9sc05668h. Chem Sci. 2020. PMID: 34123295 Free PMC article.
References
-
- Sharma V., Kumar P., Pathak D. J. Heterocycl. Chem. 2010;47:491–502.
- Ali N. A., Dar B. A., Pradhan V., Forooqui M. Mini-Rev. Med. Chem. 2013;13:1792–1800. - PubMed
-
- Jawien J., Gajda M., Rudling M., Mateuszuk L., Olszanecki R., Guzik T. J., Cichocki T., Chlopicki S., Korbut R. Eur. J. Clin. Invest. 2006;36:141–146. - PubMed
-
- Williams T. M. J. Med. Chem. 1993;36:1291–1294. - PubMed
- Silvestri R. J. Med. Chem. 2003;46:2482–2493. - PubMed
- Ragno R., Artico M., Martino G. D., Regina G. L., Coluccia A., Pasquali A. D., Silvestri R. J. Med. Chem. 2005;48:213–223. - PubMed
- Ragno R. J. Med. Chem. 2006;49:3172–3184. - PubMed
- Zhao Z. Bioorg. Med. Chem. Lett. 2008;18:554–559. - PubMed
- Famiglini V. J. Med. Chem. 2014;57:9945–9957. - PubMed
-
- De Martino G. J. Med. Chem. 2006;49:947–954. - PubMed
- La Regina G. J. Med. Chem. 2011;54:8394–8406. - PMC - PubMed
- La Regina G. J. Med. Chem. 2013;56:123–149. - PMC - PubMed
- Guan Q. Eur. J. Med. Chem. 2014;87:306–315. - PubMed
- Fortes M. P., da Silva P. B. N., da Silva T. G., Kaufman T. S., Militão G. C. G., Silveira C. C. Eur. J. Med. Chem. 2016;118:21–26. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

