Discovery of C-3 Tethered 2-oxo-benzo[1,4]oxazines as Potent Antioxidants: Bio-Inspired Based Design, Synthesis, Biological Evaluation, Cytotoxic, and in Silico Molecular Docking Studies
- PMID: 29629369
- PMCID: PMC5876303
- DOI: 10.3389/fchem.2018.00056
Discovery of C-3 Tethered 2-oxo-benzo[1,4]oxazines as Potent Antioxidants: Bio-Inspired Based Design, Synthesis, Biological Evaluation, Cytotoxic, and in Silico Molecular Docking Studies
Abstract
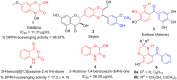
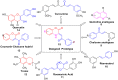
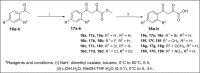
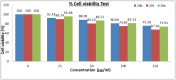
The discovery of C-3 tethered 2-oxo-benzo[1,4]oxazines as potent antioxidants is disclosed. All the analogs 20a-20ab have been synthesized via "on water" ultrasound-assisted irradiation conditions in excellent yields (upto 98%). All the compounds have been evaluated for their in vitro antioxidant activities using DPPH free radical scavenging assay as well as FRAP assay. The result showed promising antioxidant activities having IC50 values in the range of 4.74 ± 0.08 to 92.20 ± 1.54 μg/mL taking ascorbic acid (IC50 = 4.57 μg/mL) as standard reference. In this study, compounds 20b and 20t, the most active compound of the series, showed IC50 values of 6.89 ± 0.07 μg/mL and 4.74 ± 0.08 μg/mL, respectively in comparison with ascorbic acid. In addition, the detailed SAR study shows that electron-withdrawing group increases antioxidant activity and vice versa. Furthermore, in the FRAP assay, eight compounds (20c, 20j, 20m, 20n, 20r, 20u, 20z, and 20aa) were found more potent than standard reference BHT (C0.5FRAP = 546.0 ± 13.6 μM). The preliminary cytotoxic study reveals the non-toxic nature of active compounds 20b and 20t in non-cancerous 3T3 fibroblast cell lines in MTT assay up to 250 μg/mL concentration. The results were validated via carrying out in silico molecular docking studies of promising compounds 20a, 20b, and 20t in comparison with standard reference. To the best of our knowledge, this is the first detailed study of C-3 tethered 2-oxo-benzo[1,4]oxazines as potential antioxidant agents.
Keywords: 2-oxo-benzo[1,4]oxazines; BHT; DPPH; FRAP; antioxidant; ascorbic acid.
Figures



Similar articles
-
Microwave-assisted One-pot Efficient Synthesis of Functionalized 2-Oxo-2-phenylethylidenes-linked 2-Oxobenzo[1,4]oxazines and 2-Oxoquino[1,4]oxalines: Synthetic Applications, Antioxidant Activity, SAR and Cytotoxic Studies.Acta Chim Slov. 2017 Dec;64(4):988-1004. doi: 10.17344/acsi.2017.3709. Acta Chim Slov. 2017. PMID: 29318312
-
Non-peptide-based new class of platelet aggregation inhibitors: Design, synthesis, bioevaluation, SAR, and in silico studies.Arch Pharm (Weinheim). 2018 Apr;351(3-4):e1700349. doi: 10.1002/ardp.201700349. Epub 2018 Mar 9. Arch Pharm (Weinheim). 2018. PMID: 29522645
-
Synthesis, antimicrobial activity, structure-activity relationship and cytotoxic studies of a new series of functionalized (Z)-3-(2-oxo-2-substituted ethylidene)-3,4-dihydro-2H-benzo[b][1,4]oxazin-2-ones.Bioorg Med Chem Lett. 2017 Sep 15;27(18):4393-4398. doi: 10.1016/j.bmcl.2017.08.017. Epub 2017 Aug 12. Bioorg Med Chem Lett. 2017. PMID: 28835348
-
Synthesis, Bioevaluation, Structure-Activity Relationship and Docking Studies of Natural Product Inspired (Z)-3-benzylideneisobenzofuran-1(3H)-ones as Highly Potent antioxidants and Antiplatelet agents.Sci Rep. 2020 Feb 11;10(1):2307. doi: 10.1038/s41598-020-59218-6. Sci Rep. 2020. PMID: 32047204 Free PMC article.
-
Schiff's bases of quinazolinone derivatives: Synthesis and SAR studies of a novel series of potential anti-inflammatory and antioxidants.Bioorg Med Chem Lett. 2015 Mar 1;25(5):1072-7. doi: 10.1016/j.bmcl.2015.01.010. Epub 2015 Jan 13. Bioorg Med Chem Lett. 2015. PMID: 25638040 Review.
Cited by
-
Simultaneous Separation of Antioxidants and Carbohydrates From Food Wastes Using Aqueous Biphasic Systems Formed by Cholinium-Derived Ionic Liquids.Front Chem. 2019 Jun 27;7:459. doi: 10.3389/fchem.2019.00459. eCollection 2019. Front Chem. 2019. PMID: 31316969 Free PMC article.
-
Quinoline Functionalized Schiff Base Silver (I) Complexes: Interactions with Biomolecules and In Vitro Cytotoxicity, Antioxidant and Antimicrobial Activities.Molecules. 2021 Feb 24;26(5):1205. doi: 10.3390/molecules26051205. Molecules. 2021. PMID: 33668169 Free PMC article.
-
Evaluation of the Antioxidant Activity of Cis/Trans-N-Phenyl-1,4,4a,5,8,8a-Hexahydro-3,1-Benzoxazin-2-Imines.Antioxidants (Basel). 2019 Jun 25;8(6):197. doi: 10.3390/antiox8060197. Antioxidants (Basel). 2019. PMID: 31242617 Free PMC article.
-
Ag2O nanoparticle-catalyzed substrate-controlled regioselectivities: direct access to 3-ylidenephthalides and isocoumarins.RSC Adv. 2018 Jun 26;8(41):23152-23162. doi: 10.1039/c8ra03926g. eCollection 2018 Jun 21. RSC Adv. 2018. PMID: 35540153 Free PMC article.
-
MAP/Microtubule Affinity Regulating Kinase 4 Inhibitory Potential of Irisin: A New Therapeutic Strategy to Combat Cancer and Alzheimer's Disease.Int J Mol Sci. 2021 Oct 12;22(20):10986. doi: 10.3390/ijms222010986. Int J Mol Sci. 2021. PMID: 34681645 Free PMC article.
References
-
- Abdel-lateif K. S., Eldeab H. A., Maghrabi I. A. (2016). The plant natural products: their antioxidants, free radical scavengers, DNA protection and antimicrobial activities. J. Bioprocess. Biotech. 6:293 10.4172/2155-9821.1000293 - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

