Enhanced protection of C57 BL/6 vs Balb/c mice to melanoma liver metastasis is mediated by NK cells
- PMID: 29632723
- PMCID: PMC5889278
- DOI: 10.1080/2162402X.2017.1409929
Enhanced protection of C57 BL/6 vs Balb/c mice to melanoma liver metastasis is mediated by NK cells
Abstract
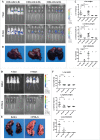
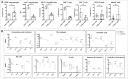
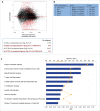
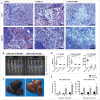
The B16F10 murine melanoma cell line displays a low expression of MHC class I molecules favoring immune evasion and metastases in immunocompetent C57 BL/6 wild-type mice. Here, we generated metastases to the liver, an organ that is skewed towards immune tolerance, by intrasplenic injection of B16F10 cells in syngeneic C57 BL/6 compared to allogeneic Balb/c mice. Surprisingly, Balb/c mice, which usually display a pronounced M2 macrophage and Th2 T cell polarization, were ∼3 times more susceptible to metastasis than C57 BL/6 mice, despite a much higher M1 and Th1 T cell immune response. The anti-metastatic advantage of C57 BL/6 mice could be attributed to a more potent NK-cell mediated cytotoxicity against B16F10 cells. Our findings highlight the role of NK cells in innate anti-tumor immunity in the context of the liver - particularly against highly aggressive MHC I-deficient cancer cells. Moreover, the B16F10 model of melanoma liver metastasis is suited for developing novel therapies targeting innate NK cell related immunity in liver metastases and liver cancer.
Keywords: NK cells; cancer immunology; innate immunity; liver; macrophages; metastasis.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
