Cepharanthine Prevents Estrogen Deficiency-Induced Bone Loss by Inhibiting Bone Resorption
- PMID: 29636680
- PMCID: PMC5880888
- DOI: 10.3389/fphar.2018.00210
Cepharanthine Prevents Estrogen Deficiency-Induced Bone Loss by Inhibiting Bone Resorption
Abstract
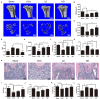
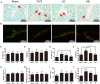
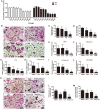
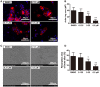
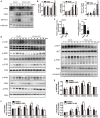
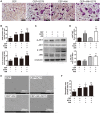
Osteoporosis is a common health problem worldwide caused by an imbalance of bone formation vs. bone resorption. However, current therapeutic approaches aimed at enhancing bone formation or suppressing bone resorption still have some limitations. In this study, we demonstrated for the first time that cepharanthine (CEP, derived from Stephania cepharantha Hayata) exerted a protective effect on estrogen deficiency-induced bone loss. This protective effect was confirmed to be achieved through inhibition of bone resorption in vivo, rather than through enhancement of bone formation in vivo. Furthermore, the in vitro study revealed that CEP attenuated receptor activator of nuclear factor κB ligand (RANKL)-induced osteoclast formation, and suppressed bone resorption by impairing the c-Jun N-terminal kinase (JNK) and phosphatidylinositol 3-kinase (PI3K)-AKT signaling pathways. The inhibitory effect of CEP could be partly reversed by treatment with anisomycin (a JNK and p38 agonist) and/or SC79 (an AKT agonist) in vitro. Our results thus indicated that CEP could prevent estrogen deficiency-induced bone loss by inhibiting osteoclastogenesis. Hence, CEP might be a novel therapeutic agent for anti-osteoporosis therapy.
Keywords: Akt; JNK; cepharanthine; osteoclasts; osteoporosis.
Figures
Similar articles
-
Leonurine hydrochloride inhibits osteoclastogenesis and prevents osteoporosis associated with estrogen deficiency by inhibiting the NF-κB and PI3K/Akt signaling pathways.Bone. 2015 Jun;75:128-37. doi: 10.1016/j.bone.2015.02.017. Epub 2015 Feb 21. Bone. 2015. PMID: 25708053
-
Boldine Ameliorates Estrogen Deficiency-Induced Bone Loss via Inhibiting Bone Resorption.Front Pharmacol. 2018 Sep 13;9:1046. doi: 10.3389/fphar.2018.01046. eCollection 2018. Front Pharmacol. 2018. PMID: 30271347 Free PMC article.
-
Caffeic acid 3,4-dihydroxy-phenethyl ester suppresses receptor activator of NF-κB ligand–induced osteoclastogenesis and prevents ovariectomy-induced bone loss through inhibition of mitogen-activated protein kinase/activator protein 1 and Ca2+–nuclear factor of activated T-cells cytoplasmic 1 signaling pathways.J Bone Miner Res. 2012 Jun;27(6):1298-1308. doi: 10.1002/jbmr.1576. J Bone Miner Res. 2012. PMID: 22337253
-
Pseurotin A Inhibits Osteoclastogenesis and Prevents Ovariectomized-Induced Bone Loss by Suppressing Reactive Oxygen Species.Theranostics. 2019 Feb 28;9(6):1634-1650. doi: 10.7150/thno.30206. eCollection 2019. Theranostics. 2019. PMID: 31037128 Free PMC article.
-
Current status and future challenges in extraction, purification and identification of Cepharanthine (a potential drug against COVID-19).Sep Purif Technol. 2023 Mar 15;309:123038. doi: 10.1016/j.seppur.2022.123038. Epub 2022 Dec 29. Sep Purif Technol. 2023. PMID: 36593875 Free PMC article. Review.
Cited by
-
Additive Effect of Parathyroid Hormone and Zoledronate Acid on Prevention Particle Wears-Induced Implant Loosening by Promoting Periprosthetic Bone Architecture and Strength in an Ovariectomized Rat Model.Front Endocrinol (Lausanne). 2022 Apr 25;13:871380. doi: 10.3389/fendo.2022.871380. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35546997 Free PMC article.
-
The brief overview, antivirus and anti-SARS-CoV-2 activity, quantitative methods, and pharmacokinetics of cepharanthine: a potential small-molecule drug against COVID-19.Front Pharmacol. 2023 Jul 31;14:1098972. doi: 10.3389/fphar.2023.1098972. eCollection 2023. Front Pharmacol. 2023. PMID: 37583901 Free PMC article. Review.
-
Dissecting the molecular mechanism of cepharanthine against COVID-19, based on a network pharmacology strategy combined with RNA-sequencing analysis, molecular docking, and molecular dynamics simulation.Comput Biol Med. 2022 Dec;151(Pt A):106298. doi: 10.1016/j.compbiomed.2022.106298. Epub 2022 Nov 11. Comput Biol Med. 2022. PMID: 36403355 Free PMC article.
-
Norcantharidin ameliorates estrogen deficient-mediated bone loss by attenuating the activation of extracellular signal-regulated kinase/ROS/NLRP3 inflammasome signaling.Front Pharmacol. 2022 Nov 3;13:1019478. doi: 10.3389/fphar.2022.1019478. eCollection 2022. Front Pharmacol. 2022. PMID: 36408264 Free PMC article.
-
A critical review: traditional uses, phytochemistry, pharmacology and toxicology of Stephania tetrandra S. Moore (Fen Fang Ji).Phytochem Rev. 2020;19(2):449-489. doi: 10.1007/s11101-020-09673-w. Epub 2020 Apr 24. Phytochem Rev. 2020. PMID: 32336965 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

