Differential Activity of the Combination of Vancomycin and Amikacin on Planktonic vs. Biofilm-Growing Staphylococcus aureus Bacteria in a Hollow Fiber Infection Model
- PMID: 29636741
- PMCID: PMC5880918
- DOI: 10.3389/fmicb.2018.00572
Differential Activity of the Combination of Vancomycin and Amikacin on Planktonic vs. Biofilm-Growing Staphylococcus aureus Bacteria in a Hollow Fiber Infection Model
Abstract
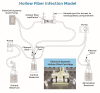
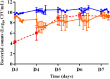
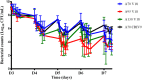
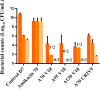
Combining currently available antibiotics to optimize their use is a promising strategy to reduce treatment failures against biofilm-associated infections. Nevertheless, most assays of such combinations have been performed in vitro on planktonic bacteria exposed to constant concentrations of antibiotics over only 24 h and the synergistic effects obtained under these conditions do not necessarily predict the behavior of chronic clinical infections associated with biofilms. To improve the predictivity of in vitro combination assays for bacterial biofilms, we first adapted a previously described Hollow-fiber (HF) infection model by allowing a Staphylococcus aureus biofilm to form before drug exposure. We then mimicked different concentration profiles of amikacin and vancomycin, similar to the free plasma concentration profiles that would be observed in patients treated daily over 5 days. We assessed the ability of the two drugs, alone or in combination, to reduce planktonic and biofilm-embedded bacterial populations, and to prevent the selection of resistance within these populations. Although neither amikacin nor vancomycin exhibited any bactericidal activity on S. aureus in monotherapy, the combination had a synergistic effect and significantly reduced the planktonic bacterial population by -3.0 to -6.0 log10 CFU/mL. In parallel, no obvious advantage of the combination, as compared to amikacin alone, was demonstrated on biofilm-embedded bacteria for which the addition of vancomycin to amikacin only conferred a further maximum reduction of 0.3 log10 CFU/mL. No resistance to vancomycin was ever found whereas a few bacteria less-susceptible to amikacin were systematically detected before treatment. These resistant bacteria, which were rapidly amplified by exposure to amikacin alone, could be maintained at a low level in the biofilm population and even suppressed in the planktonic population by adding vancomycin. In conclusion, by adapting the HF model, we were able to demonstrate the different bactericidal activities of the vancomycin and amikacin combination on planktonic and biofilm-embedded bacterial populations, suggesting that, for biofilm-associated infections, the efficacy of this combination would not be much greater than with amikacin monotherapy. However, adding vancomycin could reduce possible resistance to amikacin and provide a relevant strategy to prevent the selection of antibiotic-resistant bacteria during treatments.
Keywords: Staphylococcus aureus; amikacin; antibiotic combination; antimicrobial resistance; biofilm; hollow-fiber infection model; vancomycin.
Figures


References
-
- Adamis G., Papaioannou M. G., Giamarellos-Bourboulis E. J., Gargalianos P., Kosmidis J., Giamarellou H. (2004). Pharmacokinetic interactions of ceftazidime, imipenem and aztreonam with amikacin in healthy volunteers. Int. J. Antimicrob. Agents 23 144–149. 10.1016/j.ijantimicag.2003.07.001 - DOI - PubMed
-
- Aeschlimann J. R., Allen G. P., Hershberger E., Rybak M. J. (2000). Activities of LY333328 and vancomycin administered alone or in combination with gentamicin against three strains of vancomycin-intermediate Staphylococcus aureus in an in vitro pharmacodynamic infection model. Antimicrob. Agents Chemother. 44 2991–2998. 10.1128/AAC.44.11.2991-2998.2000 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

