TnI Structural Interface with the N-Terminal Lobe of TnC as a Determinant of Cardiac Contractility
- PMID: 29642034
- PMCID: PMC5954295
- DOI: 10.1016/j.bpj.2018.02.015
TnI Structural Interface with the N-Terminal Lobe of TnC as a Determinant of Cardiac Contractility
Abstract
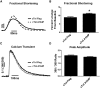
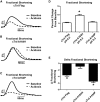
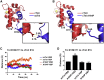
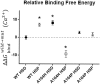
The heterotrimeric cardiac troponin complex is a key regulator of contraction and plays an essential role in conferring Ca2+ sensitivity to the sarcomere. During ischemic injury, rapidly accumulating protons acidify the myoplasm, resulting in markedly reduced Ca2+ sensitivity of the sarcomere. Unlike the adult heart, sarcomeric Ca2+ sensitivity in fetal cardiac tissue is comparatively pH insensitive. Replacement of the adult cardiac troponin I (cTnI) isoform with the fetal troponin I (ssTnI) isoform renders adult cardiac contractile machinery relatively insensitive to acidification. Alignment and functional studies have determined histidine 132 of ssTnI to be the predominant source of this pH insensitivity. Substitution of histidine at the cognate position 164 in cTnI confers the same pH insensitivity to adult cardiac myocytes. An alanine at position 164 of cTnI is conserved in all mammals, with the exception of the platypus, which expresses a proline. Prolines are biophysically unique because of their innate conformational rigidity and helix-disrupting function. To provide deeper structure-function insight into the role of the TnC-TnI interface in determining contractility, we employed a live-cell approach alongside molecular dynamics simulations to ascertain the chemo-mechanical implications of the disrupted helix 4 of cTnI where position 164 exists. This important motif belongs to the critical switch region of cTnI. Substitution of a proline at position 164 of cTnI in adult rat cardiac myocytes causes increased contractility independent of alterations in the Ca2+ transient. Free-energy perturbation calculations of cTnC-Ca2+ binding indicate no difference in cTnC-Ca2+ affinity. Rather, we propose the enhanced contractility is derived from new salt bridge interactions between cTnI helix 4 and cTnC helix A, which are critical in determining pH sensitivity and contractility. Molecular dynamics simulations demonstrate that cTnI A164P structurally phenocopies ssTnI under baseline but not acidotic conditions. These findings highlight the evolutionarily directed role of the TnI-cTnC interface in determining cardiac contractility.
Copyright © 2018. Published by Elsevier Inc.
Figures



Similar articles
-
Molecular determinants of cardiac myocyte performance as conferred by isoform-specific TnI residues.Biophys J. 2014 May 20;106(10):2105-14. doi: 10.1016/j.bpj.2014.04.017. Biophys J. 2014. PMID: 24853739 Free PMC article.
-
Ca(2+) induces an extended conformation of the inhibitory region of troponin I in cardiac muscle troponin.J Mol Biol. 2001 Nov 16;314(1):51-61. doi: 10.1006/jmbi.2001.5118. J Mol Biol. 2001. PMID: 11724531
-
Solution structures of the C-terminal domain of cardiac troponin C free and bound to the N-terminal domain of cardiac troponin I.Biochemistry. 1999 Jun 29;38(26):8313-22. doi: 10.1021/bi9902642. Biochemistry. 1999. PMID: 10387077
-
Cardiac troponin structure-function and the influence of hypertrophic cardiomyopathy associated mutations on modulation of contractility.Arch Biochem Biophys. 2016 Jul 1;601:11-21. doi: 10.1016/j.abb.2016.02.004. Epub 2016 Feb 4. Arch Biochem Biophys. 2016. PMID: 26851561 Free PMC article. Review.
-
Focus on cardiac troponin complex: From gene expression to cardiomyopathy.Genes Dis. 2024 Mar 11;11(6):101263. doi: 10.1016/j.gendis.2024.101263. eCollection 2024 Nov. Genes Dis. 2024. PMID: 39211905 Free PMC article. Review.
Cited by
-
Prognostic value of prealbumin, N-terminal pro-B-type natriuretic peptide, heart type fatty acid binding protein, and cardiac troponin I in elderly patients for heart failure and poor outcomes.J Int Med Res. 2021 May;49(5):300060521999742. doi: 10.1177/0300060521999742. J Int Med Res. 2021. PMID: 34039072 Free PMC article.
-
Loss of dysferlin or myoferlin results in differential defects in excitation-contraction coupling in mouse skeletal muscle.Sci Rep. 2021 Aug 5;11(1):15865. doi: 10.1038/s41598-021-95378-9. Sci Rep. 2021. PMID: 34354129 Free PMC article.
-
OR-1896 increases force of contraction in the isolated human atrium.Naunyn Schmiedebergs Arch Pharmacol. 2023 Dec;396(12):3823-3833. doi: 10.1007/s00210-023-02592-5. Epub 2023 Jun 24. Naunyn Schmiedebergs Arch Pharmacol. 2023. PMID: 37354216 Free PMC article.
-
Computational Studies of Cardiac and Skeletal Troponin.Front Mol Biosci. 2019 Aug 9;6:68. doi: 10.3389/fmolb.2019.00068. eCollection 2019. Front Mol Biosci. 2019. PMID: 31448287 Free PMC article. Review.
-
Levosimendan increases the phosphorylation state of phospholamban in the isolated human atrium.Naunyn Schmiedebergs Arch Pharmacol. 2023 Apr;396(4):669-682. doi: 10.1007/s00210-022-02348-7. Epub 2022 Nov 29. Naunyn Schmiedebergs Arch Pharmacol. 2023. PMID: 36445386 Free PMC article.
References
-
- Huxley H., Hanson J. Changes in the cross-striations of muscle during contraction and stretch and their structural interpretation. Nature. 1954;173:973–976. - PubMed
-
- Rayment I., Rypniewski W.R., Holden H.M. Three-dimensional structure of myosin subfragment-1: a molecular motor. Science. 1993;261:50–58. - PubMed
-
- Granzier H.L., Labeit S. The giant protein titin: a major player in myocardial mechanics, signaling, and disease. Circ. Res. 2004;94:284–295. - PubMed
-
- Thompson B.R., Asp M.L., Metzger J.M. Molecular mechanism of sarcomeric cardiomyopathies. In: Garry D., Wilson R., Vlodaver Z., editors. Congestive Heart Failure and Cardiac Transplantation. Springer International Publishing; 2017. pp. 151–160.
-
- Farah C.S., Reinach F.C. The troponin complex and regulation of muscle contraction. FASEB J. 1995;9:755–767. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

